Acromegaloid Facial Appearance Syndrome Treatments
Find Acromegaloid Facial Appearance Syndrome Treatments
Mycapssa
What is Mycapssa (Octreotide)?
Approved To Treat
Related Clinical Trials
Summary: Gastro-enteropancreatic Neuro-endocrine tumors (GEP-NETs) are rare slow-growing cancers which commonly involve the abdominal organs like liver, stomach, intestines and pancreas. Their incidence has been documented to have increased over the last decade, thus resulting in treatment options being developed to treat these cancers. These cancers spread commonly to the liver, followed by lungs, bones a...
Summary: The purpose of the current study is to evaluate the efficacy and safety of \[177Lu\]Lu-DOTA-TATE plus octreotide long-acting release (LAR) versus octreotide LAR alone in newly diagnosed patients with somatostatin receptor positive (SSTR+), well differentiated Grade1 and Grade 2 (G1 and G2) (Ki-67 \<10%) advanced gastroenteropancreatic neuroendocrine tumors (GEP-NETs) with high disease burden
Summary: The primary purpose of this study is to assess the effect of Debio 4126 in the maintenance of the levels of insulin-like growth factor 1 (IGF-1) ≤1x upper limit of normal (ULN) in the double-blind period (Period 1) in comparison to placebo at week 36.
Related Latest Advances
Brand Information
- Cholelithiasis and Complications of Cholelithiasis
- Hyperglycemia and Hypoglycemia
- Thyroid Function Abnormalities
- Cardiac Function Abnormalities
- Steatorrhea and Malabsorption of Dieatary Fats
- Changes in Vitamin B
- Blood and lymphatic: pancytopenia, thrombocytopenia
- Cardiac: myocardial infarction, cardiac arrest, atrial fibrillation
- Ear and labyrinth: deafness
- Endocrine: diabetes insipidus, adrenal insufficiency in patients 18 months of age and under, pituitary apoplexy
- Eye: glaucoma, visual field defect, scotoma, retinal vein thrombosis
- Gastrointestinal: intestinal obstruction, peptic/gastric ulcer, abdomen enlarged
- General and administration site: generalized edema, facial edema
- Hepatobiliary: gallbladder polyp, fatty liver, hepatitis
- Immune: anaphylactoid reactions including anaphylactic shock
- Infections and infestations: appendicitis
- Laboratory abnormalities: increased liver enzymes, CK increased, creatinine increased
- Metabolism and nutrition: diabetes mellitus
- Musculoskeletal: arthritis, joint effusion, Raynaud's syndrome
- Nervous System: convulsions, aneurysm, intracranial hemorrhage, hemiparesis, paresis, suicide attempt, paranoia, migraines, Bell's palsy, aphasia
- Renal and urinary: renal failure, renal insufficiency
- Reproductive and breast: gynecomastia, galactorrhea, libido decrease, breast carcinoma
- Respiratory: status asthmaticus, pulmonary hypertension, pulmonary nodule, pneumothorax aggravated
- Skin and subcutaneous tissue: urticaria, cellulitis, petechiae
- Vascular: orthostatic hypotension, hematuria, gastrointestinal hemorrhage, arterial thrombosis of the arm
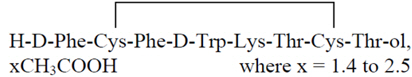
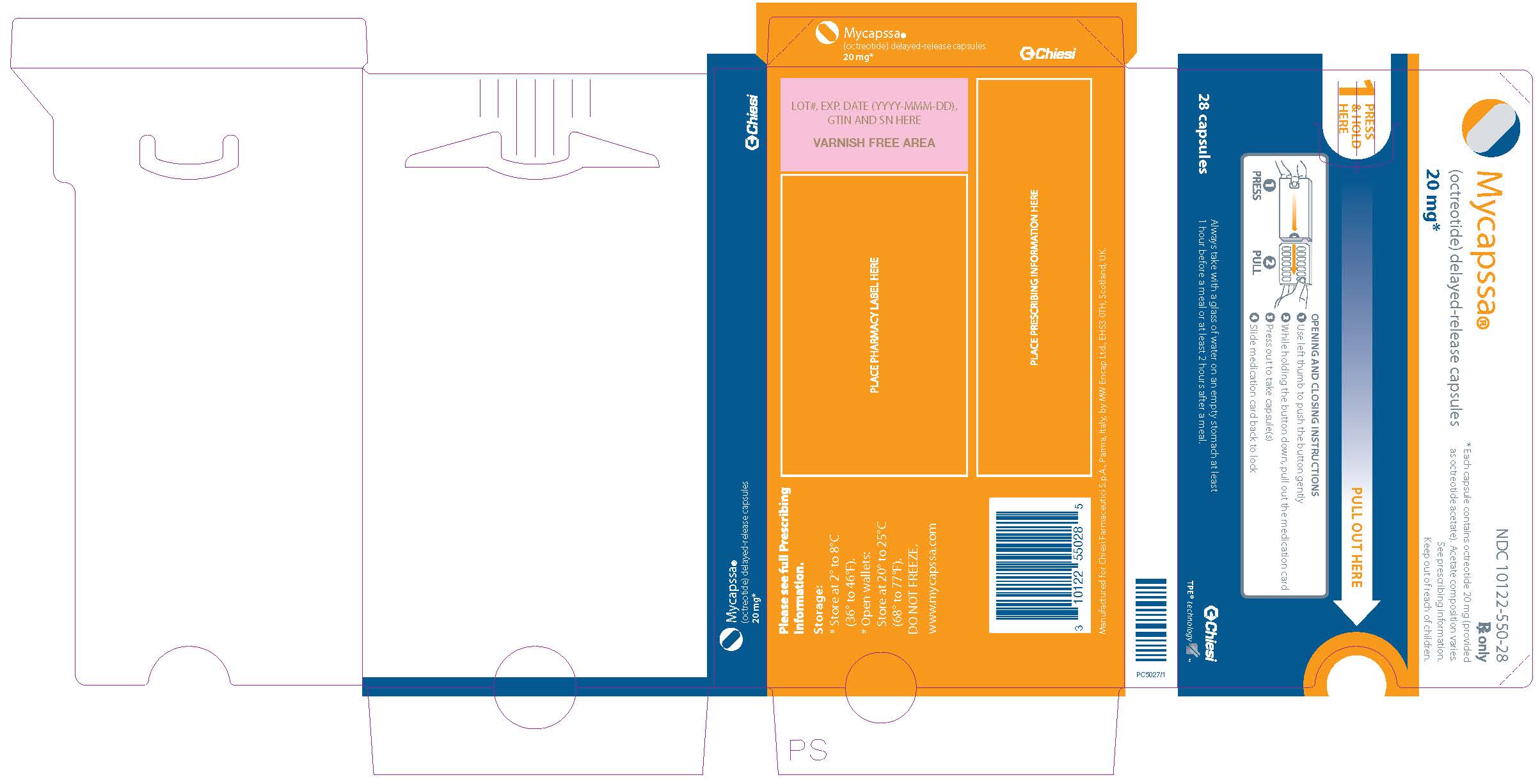
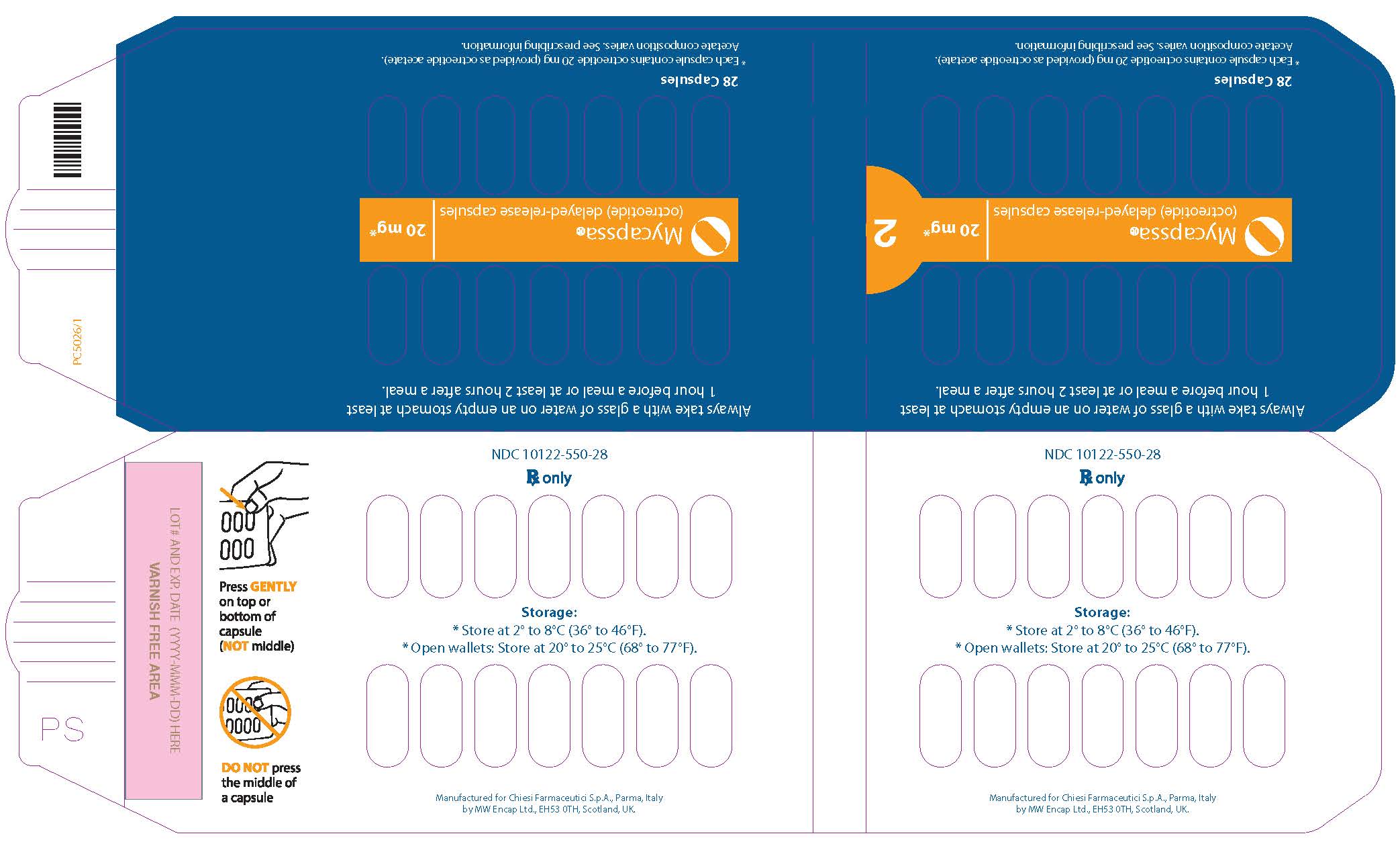
- Each MYCAPSSA wallet contains twenty-eight 20-mg capsules. The number of wallets required in a 28-day period depends on your prescribed dose.
- Each MYCAPSSA wallet has a locking mechanism that helps to keep the medicine away from children.
- Become familiar with using the MYCAPSSA wallet so you will know how to use it the right way.
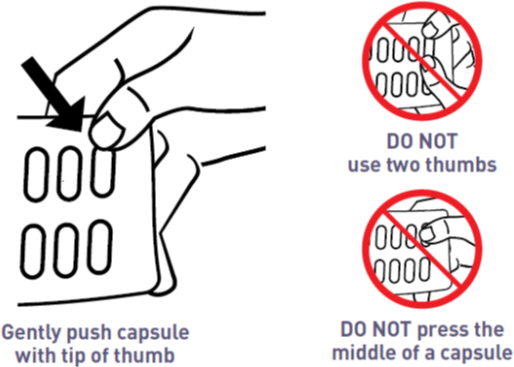
- Place the tip of a thumb at the edge of a capsule's plastic cavity (see
- Gently push the capsule until it is removed. Collect the removed capsule in your hand.
- Do not use two thumbs to push a capsule as this could damage it.
- Do not press the middle of a capsule. This could also damage it.
- If a capsule is cracked or broken, throw it away (discard it) and remove another capsule.
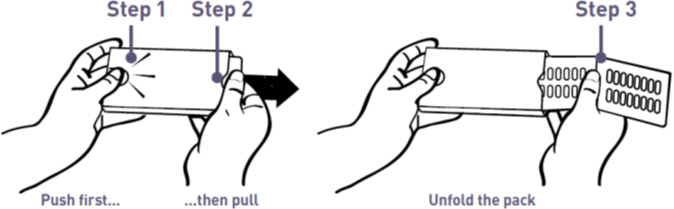
- Use left thumb to push the button gently
- While holding the button down, pull out the medication card
- Press out to take capsule(s)
- Slide medication card back to lock