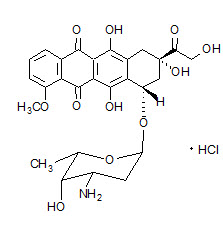
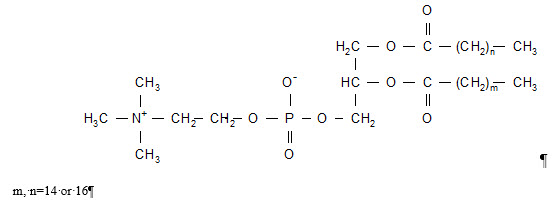
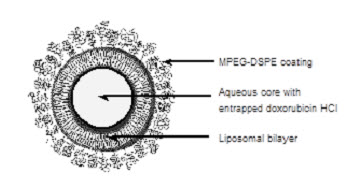
Doxil
What is Doxil (DOXOrubicin)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: This pilot trial assesses the effect of the combination of blinatumomab with dasatinib or imatinib and standard chemotherapy for treating patients with Philadelphia chromosome positive (Ph+) or ABL-class Philadelphia chromosome-like (Ph-like) B-Cell acute lymphoblastic leukemia (B-ALL). Blinatumomab is a bispecific antibody that binds to two different proteins-one on the surface of cancer cells an...
Summary: This phase I trial tests the safety, side effects, and best dose of combination therapy with liposomal doxorubicin and peposertib in treating patients with sarcoma that has spread from where it first started, to other places in the body (metastatic), or cannot be removed by surgery (unresectable) and for which no known cure is available (advanced). Doxorubicin is in a class of medications called a...
Summary: This phase III trial compares the effect of adding immunotherapy (brentuximab vedotin and nivolumab) to standard treatment (chemotherapy with or without radiation) to the standard treatment alone in improving survival in patients with stage I and II classical Hodgkin lymphoma. Brentuximab vedotin is in a class of medications called antibody-drug conjugates. It is made of a monoclonal antibody call...
Related Latest Advances
Brand Information
- DOXIL liposomal infusion can cause myocardial damage, including acute left ventricular failure. The risk of cardiomyopathy was 11% when the cumulative anthracycline dose was between 450 mg/m
- Serious, life-threatening, and fatal infusion-related reactions can occur with DOXIL liposomal infusion Acute infusion-related reactions occurred in 11% of patients with solid tumors. Withhold DOXIL liposomal infusion for infusion-related reactions and resume at a reduced rate. Discontinue DOXIL liposomal infusion for serious or life-threatening infusion-related reactions
- Cardiomyopathy
- Infusion-Related Reactions
- Hand-Foot Syndrome
- Secondary Oral Neoplasms
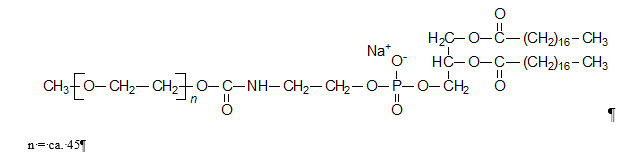
- “Hazardous Drugs”,
