Chronic Obstructive Pulmonary Disease (COPD) Treatments
Find Chronic Obstructive Pulmonary Disease (COPD) Treatments
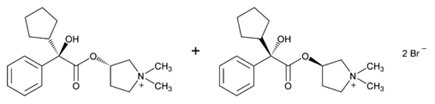
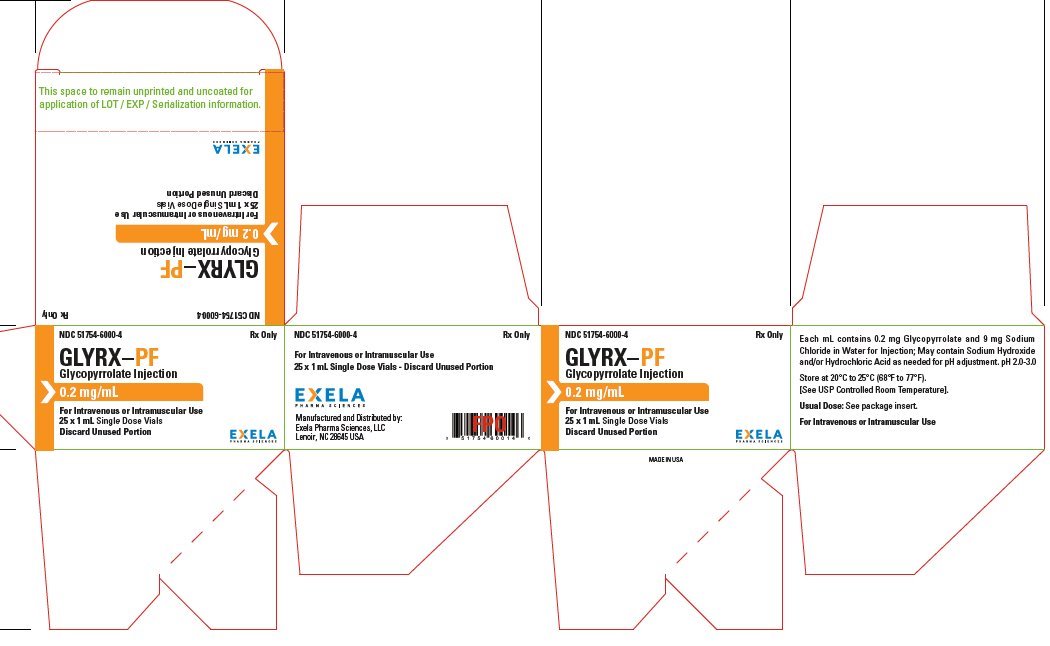
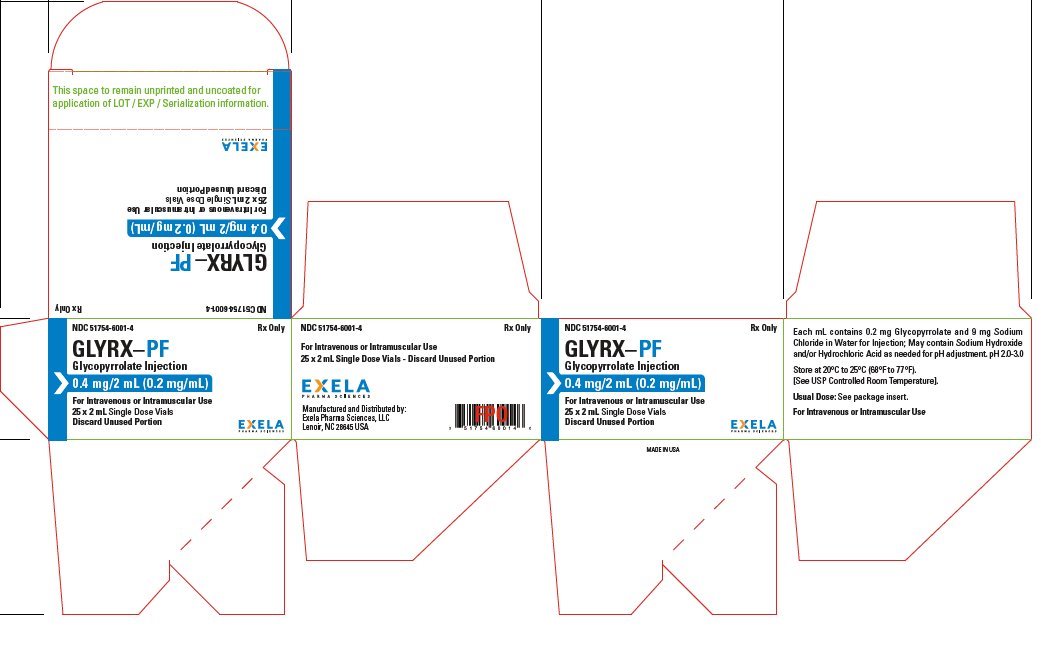
Glycopyrrolate
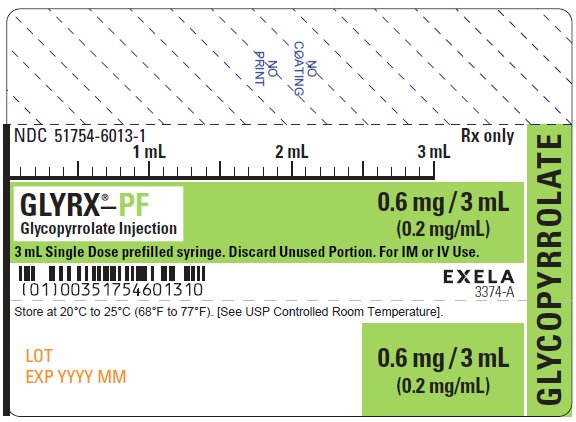
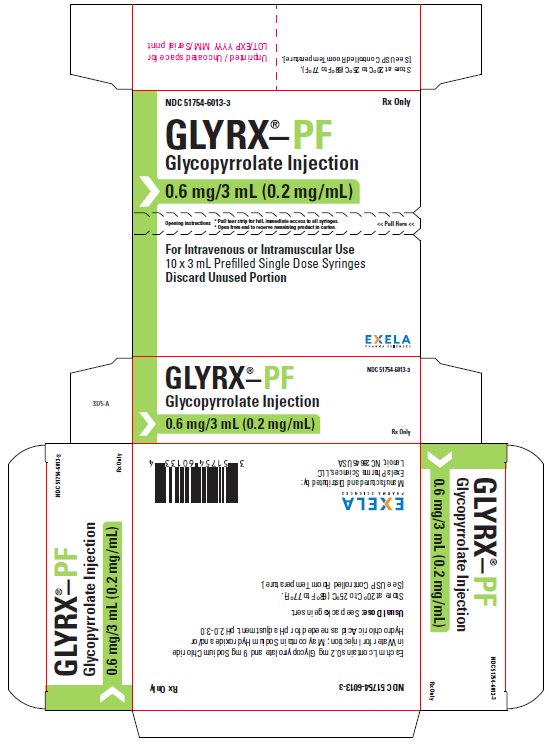
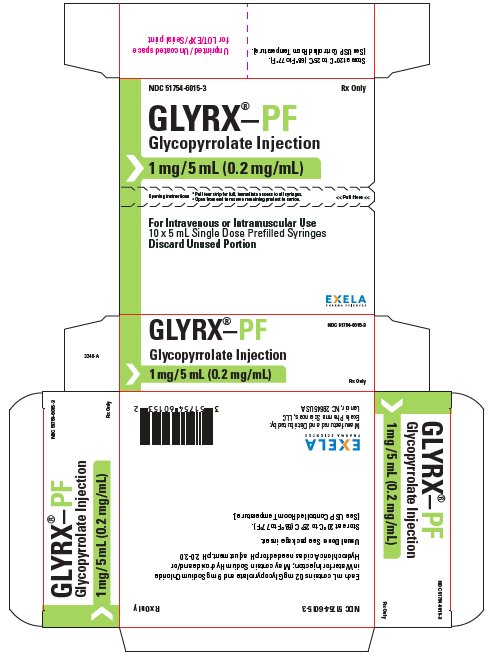
What is Glyrx-Pf (Glycopyrrolate)?
Top Global Experts
Related Clinical Trials
Summary: This study will assess the safety and efficacy of fixed dose combinations of ensifentrine with two different glycopyrrolate dose levels compared to placebo and to the individual components of the fixed dose combinations, each administered twice a day via standard jet nebulizer, in adult subjects with chronic obstructive pulmonary disease (COPD).
Summary: The purpose of this study is to characterize the bronchodilator effect, systemic exposure and safety/tolerability of two different doses of inhaled glycopyrronium, when compared to placebo. Outcome of this study will be used to determine the dose of inhaled glycopyrronium for the development of fixed dose combination indacaterol/mometasone/glycopyrronium (QVM149) for children aged 6 to less than 1...
Summary: The CHOROS pooled analysis is a retrospective secondary data use analysis of integrated individual participant data from a series of planned and on-going primary prospective, non-interventional, multi-center studies sponsored by AstraZeneca and conducted in the pulmonary/primary care practitioner setting in multiple countries and may include data from the following countries: United Kingdom, Germa...
Related Latest Advances
Brand Information
- for reduction of salivary, tracheobronchial, and pharyngeal secretions, reduction of volume and acidity of gastric secretions, and blockade of cardiac inhibitory reflexes during induction of anesthesia and intubation,
- intraoperatively to counteract surgically or drug-induced or vagal reflex-associated arrhythmias, and
- for protection against peripheral muscarinic effects of cholinergic agents such as neostigmine and pyridostigmine given to reverse the neuromuscular blockade due to non-depolarizing agents.
- To reduce symptoms of a peptic ulcer as an adjunct to treatment of peptic ulcer when rapid anticholinergic effect is desired or when oral medication is not tolerated.
- Limitations of Use
GLYRX-PF is not indicated as monotherapy for the treatment of peptic ulcer because effectiveness in peptic ulcer healing has not been established.
- patients with known hypersensitivity to glycopyrrolate or any of its inactive ingredients.
- peptic ulcer patients with the following concurrent conditions: glaucoma; obstructive uropathy (for example, bladder neck obstruction due to prostatic hypertrophy); obstructive disease of the gastrointestinal tract (as in achalasia, pyloroduodenal stenosis, etc.); paralytic ileus, intestinal atony of the elderly or debilitated patient; unstable cardiovascular status in acute hemorrhage; severe ulcerative colitis; toxic megacolon complicating ulcerative colitis; myasthenia gravis.