Heartburn Treatments
Find Heartburn Treatments
Metoclopramide
What is Reglan (Metoclopramide)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: Metoclopramide is a drug approved by the FDA for gastroesophageal reflux and to relieve symptoms in adults with acute and recurrent diabetic gastroparesis. The objective of this study is to determine whether metoclopramide can improve hypoglycemia awareness and decrease the incidence of hypoglycemia in type 1 diabetes patients with hypoglycemia unawareness.
Summary: The goal of this clinical trial is to evaluate the efficacy and safety of olanzapine plus metoclopramide in preventing opioid-induced nausea and vomiting (OINV) in adult patients with advanced cancer who are initiating strong opioid therapy. The main questions it aims to answer are: (1) Does the combination of olanzapine and metoclopramide reduce the incidence of OINV? (2)What adverse events do pa...
Summary: The goal of this clinical trial is to examine immune responses to the BCG vaccine in healthy adults who have, or who have not, taken antibiotics to deplete their gut bacteria prior to vaccination. The main question it aims to answer is: does depletion of the gut microbiota lead to impaired BCG-induced protection against specific and non-specific to challenges to the immune system?
Related Latest Advances
Brand Information
- Metoclopramide, including Reglan, can cause tardive dyskinesia (TD), a potentially irreversible serious movement disorder. In patients treated with metoclopramide, including Reglan, the risk of developing TD increases with duration of treatment and total cumulative dosage
- Reglan is contraindicated in patients with a history of TD.
- Use Reglan for the shortest duration of treatment and periodically reassess the need for continued treatment.
- Immediately discontinue Reglan in patients who develop signs or symptoms of TD
- In patients with symptomatic, documented gastroesophageal reflux, the maximum duration of Reglan treatment is 12 weeks
- In patients with diabetic gastroparesis, avoid a total duration of treatment with metoclopramide products, including Reglan tablets, for longer than 12 weeks. If longer term use is unavoidable, routinely monitor for signs and symptoms of TD
- Treatment for 4 to 12 weeks of symptomatic, documented gastroesophageal reflux in adults who fail to respond to conventional therapy.
- Relief of symptoms in adults with acute and recurrent diabetic gastroparesis.
- Reglan has not been shown to be safe and effective for the treatment of symptomatic, documented gastroesophageal reflux for longer than 12 weeks
- Reglan tablets are not recommended for use in pediatric patients due to the risk of developing tardive dyskinesia (TD) and other extrapyramidal symptoms as well as the risk of methemoglobinemia in neonates
- 5 mg metoclopramide: green, elliptical-shaped, debossed “REGLAN” over “5” on one side and “ANI” on the opposite side
- 10 mg metoclopramide: white, double edge scored, capsule-shaped, debossed “REGLAN” on one side and “ANI 10” on the opposite side
- In patients with a history of tardive dyskinesia (TD) or a dystonic reaction to metoclopramide
- When stimulation of gastrointestinal motility might be dangerous (e.g., in the presence of gastrointestinal hemorrhage, mechanical obstruction, or perforation).
- In patients with pheochromocytoma or other catecholamine-releasing paragangliomas. Reglan may cause a hypertensive/pheochromocytoma crisis, probably due to release of catecholamines from the tumor
- In patients with epilepsy. Reglan may increase the frequency and severity of seizures
- In patients with hypersensitivity to metoclopramide. Reactions have included laryngeal and glossal angioedema and bronchospasm
- Tardive dyskinesia
- Other extrapyramidal symptoms
- Neuroleptic malignant syndrome
- Depression
- Hypertension
- Fluid retention
- Hyperprolactinemia
- Effects on the ability to drive and operate machinery
- Tardive dyskinesia, acute dystonic reactions, drug-induced parkinsonism, akathisia, and other extrapyramidal symptoms
- Convulsive seizures
- Hallucinations
- Restlessness, drowsiness, fatigue, and lassitude occurred in approximately 10% of patients who received 10 mg four times daily. Insomnia, headache, confusion, dizziness, or depression with suicidal ideation occurred less frequently.
- Neuroleptic malignant syndrome, serotonin syndrome (in combination with serotonergic agents).
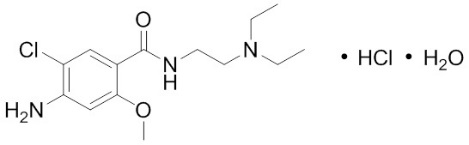
- Each Reglan 5 mg tablet contains 5 mg metoclopramide (equivalent to 5.91 mg of metoclopramide hydrochloride USP). Inactive ingredients consist of corn starch, D&C Yellow 10 Aluminum Lake, FD&C Blue 1 Aluminum Lake, lactose, microcrystalline cellulose, silicon dioxide, and stearic acid.
- Each Reglan 10 mg tablet contains 10 mg metoclopramide (equivalent to 11.82 mg metoclopramide hydrochloride USP). Inactive ingredients consist of magnesium stearate, mannitol, microcrystalline cellulose, and stearic acid.

