Hepatitis B Treatments
Find Hepatitis B Treatments
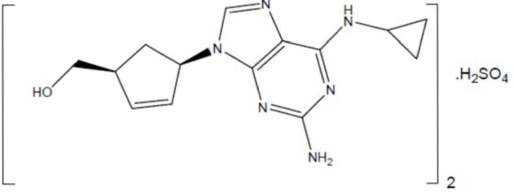
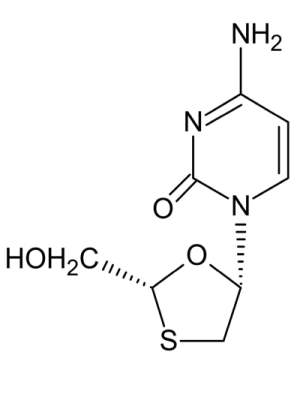
Lamivudine
What is Abacavir (Lamivudine)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: The study investigators are conducting foundational pharmacokinetic (PK) and qualitative studies, among 15-24 years old (inclusive) adolescent girls and young women living with HIV (AGYWLHIV) already on oral antiretroviral therapy (ART) and virally suppressed, leading up to a hybrid type I effectiveness-implementation trial randomizing individual AGYWLHIV to receive long-acting (LA) injectable cab...
Background: Lymphoma is a type of blood cancer. Blood cell transplant can cure some people with lymphoma. Researchers want to see if they can limit the complications transplant can cause.
Summary: Treatment adherence is defined by compliance with the dosage schedule (i.e., dose per dose and number of doses per day), as well as the duration of administration (i.e., number of days during which the dosage schedule must be followed). Treatment adherence determines the therapeutic efficacy and the absence of toxicity of the prescribed medication. However, this adherence is far from being respect...
Related Latest Advances
Brand Information
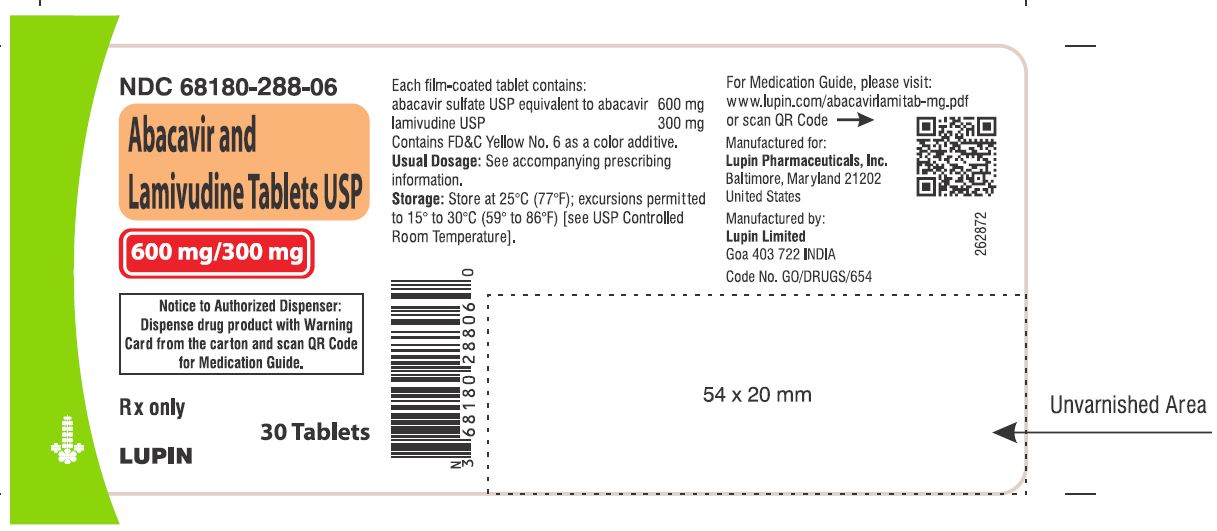
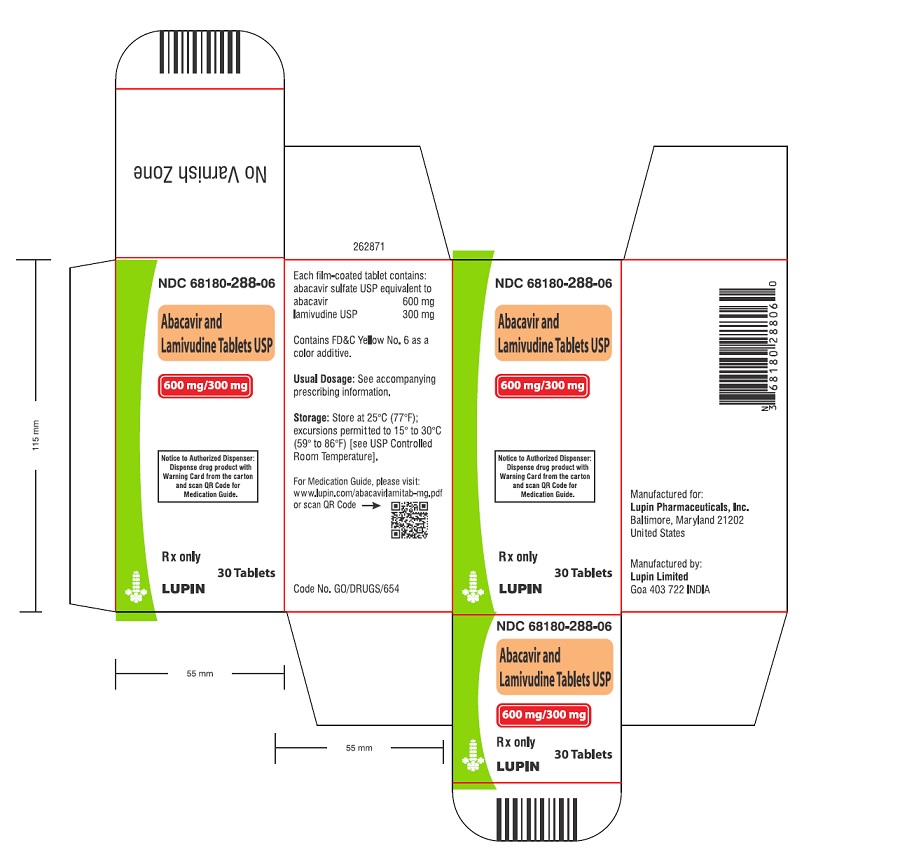
- that a Medication Guide and Warning Card summarizing the symptoms of the abacavir hypersensitivity reaction and other product information will be dispensed by the pharmacist with each new prescription and refill of abacavir and lamivudine tablets, and instruct the patient to read the Medication Guide and Warning Card every time to obtain any new information that may be present about abacavir and lamivudine tablets. The complete text of the Medication Guide is reprinted at the end of this document.
- to carry the Warning Card with them.
- how to identify a hypersensitivity reaction
- that if they develop symptoms consistent with a hypersensitivity reaction they should call their healthcare provider right away to determine if they should stop taking abacavir and lamivudine tablets.
- that a hypersensitivity reaction can worsen and lead to hospitalization or death if abacavir and lamivudine tablets is not immediately discontinued.
- to not restart abacavir and lamivudine tablets or any other abacavir-containing product following a hypersensitivity reaction because more severe symptoms can occur within hours and may include life-threatening hypotension and death.
- that if they have a hypersensitivity reaction, they should dispose of any unused abacavir and lamivudine tablets to avoid restarting abacavir.
- that a hypersensitivity reaction is usually reversible if it is detected promptly and abacavir and lamivudine tablets is stopped right away.
- that if they have interrupted abacavir and lamivudine tablets for reasons other than symptoms of hypersensitivity (for example, those who have an interruption in drug supply), a serious or fatal hypersensitivity reaction may occur with reintroduction of abacavir.
- to not restart abacavir and lamivudine tablets or any other abacavir-containing product without medical consultation and only if medical care can be readily accessed by the patient or others.