Hypoparathyroidism Treatments
Find Hypoparathyroidism Treatments
Vectical
What is Vectical (Calcitriol)?
Approved To Treat
Related Clinical Trials
Summary: Orthodontic treatment (OT) is characterized as a lengthy procedure and enormous effort has been harnessed to shorten the OT duration using either surgical or non-surgical approaches. With the emerging concept of accelerated orthodontics, scientific interest has been focused on chemical and electrical stimuli in combination with mechanical forces for rapid bone turnover and more stable results. Giv...
Summary: Alopecia areata (AA) is an immunologically mediated disease characterized by non-scarring hair loss (Madni and Shapiro, 2000). AA is presented by rapid and complete hair loss in one or more round or oval patches, usually on the scalp, beard area, eyebrows, eyelashes, and less commonly, on other hairy areas of the body (Berker et al., 2010 and James et al., 2011) Alopecia areata is a T-cell-mediate...
Summary: This study aims to compare two different protocols commonly used in the management of post thyroidectomy hypoparathyroidism : PTH based calcium repletion and empiric repletion. The investigators aim to compare the quality of life associated with these two protocols in a randomized trial.
Related Latest Advances
Brand Information
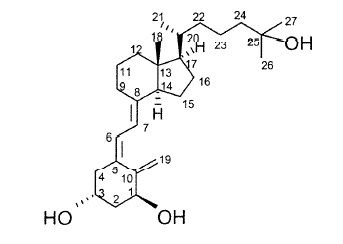
Pediatric Patients
The systemic exposure of calcitriol was assessed in pediatric subjects ages 2 to 17 years with plaque psoriasis in two trials. In one trial, 25 subjects ages 12 to 17 applied calcitriol ointment 3 mcg/g twice a day for 8 weeks to a body surface area of 10% to 35%. The mean daily dose was 10.43 g/day. In the second trial, 17 subjects ages 2 to 12 applied calcitriol ointment 3 mcg/g twice a day for 14 days to a body surface area of 3% to 18%. The mean daily dose was 17.09 g/day. In both trials, the systemic concentrations of calcitriol post treatment were relatively flat and were generally comparable to the endogenous levels observed at baseline. The PK parameters could not be reliably estimated.
There was no correlation between the elevated calcitriol levels and the pharmacodynamic parameters of serum albumin adjusted calcium, serum phosphorus, urinary calcium and urinary phosphorus.
- This medication is to be used as directed by the physician. It is for external use only. This medication is to be applied only to areas of the skin affected by psoriasis, as directed. It should be gently rubbed into the skin so that no medication remains visible.
- This medication may affect calcium metabolism. Hypercalcemia has been observed in subjects exposed to this medicine. Increased absorption may occur with use of occlusive dressings.
- Avoid use of more than 100 grams per week in patients ages 2-6 years and use of more than 200 grams per week in patients ages 7 years and older.
- Instruct patients to report any signs of adverse reactions to their physician.
- Avoid contact with eyes, lips, and facial skin.
- Advise breastfeeding women not to apply VECTICAL Ointment directly to the nipple and areola to avoid direct infant exposure

GALDERMA
Each gram contains:calcitriol 3mcg in an ointment base consisting of mineral oil, dl-α-tocopherol, and white petrolatum,
Do not freeze or refrigerate.
Marketed by: GALDERMA LABORATORIES, L.P.
Dallas, Texas 75201 USA
Made in Canada.
All trademarks are the property of their respective owners.
P51459-6
