Interstitial Lung Disease Treatments
Find Interstitial Lung Disease Treatments
Esbriet
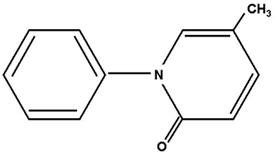
What is Esbriet (Pirfenidone)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: This study is open to adults with idiopathic pulmonary fibrosis (IPF) who are 40 years and older. The purpose of this study is to find out whether a medicine called pirfenidone changes the amount of a medicine called BI 1015550 in the blood. Some people may take more than one medicine at a time. Therefore, it is important to understand how different medicines influence one another. Participants ta...
Summary: A randomized, double-blind, placebo-controlled clinical study to evaluate the safety and efficacy of 2 doses of inhaled pirfenidone (AP01) versus placebo on top of standard of care in participants with PPF over 52 weeks.
Summary: This study will evaluate the impact Nintedanib Solution for Inhalation (AP02) has on lung function and key measures of fibrosis in adult patients with idiopathic pulmonary fibrosis (IPF) as well as assess its safety and tolerability. Adults 40 years of age or older with IPF who meet the inclusion and exclusion criteria can participate in this study if they are not currently on treatment for IPF, a...
Related Latest Advances
Brand Information
- Liver Enzyme Elevations and Drug-Induced Liver Injury
- Photosensitivity Reaction or Rash
- Severe Cutaneous Adverse Reactions
- Gastrointestinal Disorders
- NDC 50242-121-01, bottle for a 30-day supply containing 270 capsules and closed with a child-resistant closure
- NDC 50242-121-02, 14-day titration blister pack, carton containing a total of 63 capsules in two blister cards – a Week 1 blister card containing 21 capsules (1 capsule per blister well) and a Week 2 blister card containing 42 capsules (2 capsules per blister well)
- NDC 50242-121-03, 4-week maintenance blister pack, carton containing a total of 252 capsules in four blister cards each with 63 capsules (3 capsules per blister well)
- NDC 50242-122-05, carton containing 3 bottles, each containing ninety 267 mg tablets (270 tablets total) with a child-resistant closure
- NDC 50242-122-06, carton containing 1 bottle containing 270 tablets, 267 mg each, with a child-resistant closure
- NDC 50242-123-01, carton containing 1 bottle containing ninety 801 mg tablets, with a child-resistant closure


(pirfenidone)
tablets
90 tablets





