Sinusitis Treatments
Find Sinusitis Treatments
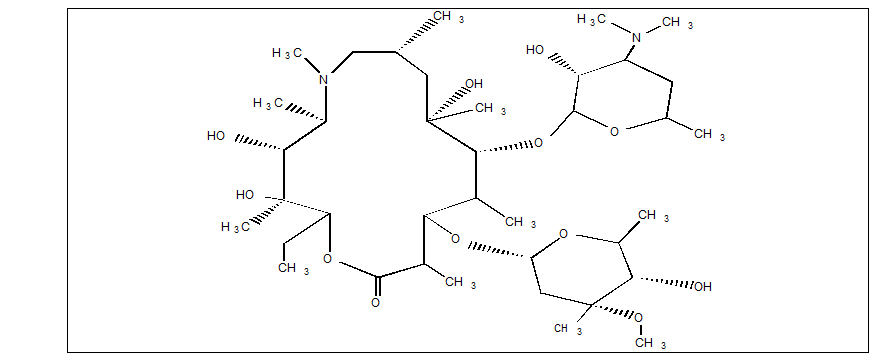
Azithromycin
What is Zithromax (Azithromycin)?
Top Global Experts
Related Clinical Trials
Summary: This double-blind, randomized, placebo-controlled, multinational, multicenter, parallel-group, Phase 3, 2-arm, study will investigate the efficacy and safety of belumosudil compared with placebo, both administered on top of azithromycin and standard-of-care regimen of immunosuppression in male or female participants at least 1 year after bilateral lung transplant, who are at least 18 years of age ...
Summary: This is a phase-III multi-center double-blind randomized controlled trial of 8,000 individuals undergoing a scheduled or prelabor cesarean delivery who are randomized to either adjunctive azithromycin prophylaxis or to placebo. Both groups also will receive standard of care preoperative antibiotics (excluding azithromycin). The primary endpoint is a maternal infection composite defined as any one ...
Summary: The study investigators are interested in learning more about how drugs, that are given to children by their health care provider, act in the bodies of children and young adults in hopes to find the most safe and effective dose for children. The primary objective of this study is to evaluate the PK of understudied drugs currently being administered to children per SOC as prescribed by their treati...
Related Latest Advances
Brand Information
- Hypersensitivity
- Hepatotoxicity
- Infantile Hypertrophic Pyloric Stenosis (IHPS)
- QT Prolongation
- Cardiovascular Death
- Clostridioides difficile-Associated Diarrhea (CDAD) [see
- Exacerbation of Myasthenia Gravis
Nervous system: Headache, somnolence.
Allergic: Bronchospasm.
Special senses: Taste perversion.
Cardiovascular: Arrhythmias including ventricular tachycardia and hypotension. There have been reports of QT prolongation, torsades de pointes, and cardiovascular death.
Gastrointestinal: Anorexia, constipation, dyspepsia, flatulence, vomiting/diarrhea, pseudomembranous colitis, pancreatitis, oral candidiasis, pyloric stenosis, and reports of tongue discoloration.
General: Asthenia, paresthesia, fatigue, malaise and anaphylaxis (including fatalities).
Genitourinary: Interstitial nephritis and acute renal failure and vaginitis.
Hematopoietic: Thrombocytopenia.
Liver/biliary: Abnormal liver function, hepatitis, cholestatic jaundice, hepatic necrosis, and hepatic failure. [see
Nervous system: Convulsions, dizziness/vertigo, headache, somnolence, hyperactivity, nervousness, agitation and syncope.
Psychiatric: Aggressive reaction and anxiety.
Skin/appendages: Pruritus, serious skin reactions including, erythema multiforme, AGEP, Stevens-Johnson syndrome, toxic epidermal necrolysis, and DRESS.
Special senses: Hearing disturbances including hearing loss, deafness and/or tinnitus and reports of taste/smell perversion and/or loss.
- elevated ALT (SGPT), AST (SGOT), creatinine (4 to 6%)
- elevated LDH, bilirubin (1 to 3%)
- leukopenia, neutropenia, decreased platelet count, and elevated serum alkaline phosphatase (less than 1%)