Swimmer's Ear Treatments
Find Swimmer's Ear Treatments
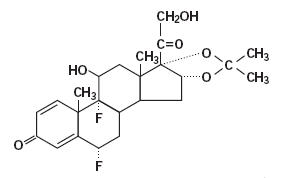
Fluocinolone Acetonide
What is Synalar (Fluocinolone Acetonide)?
Approved To Treat
Top Global Experts
There are no experts for this drug
Related Clinical Trials
Summary: This randomized controlled trial will evaluate the effect of intravitreal faricimab or fluocinolone acetonide (FAc) intravitreal implant compared with observation on long-term visual acuity following treatment of choroidal melanoma with iodine-125 plaque brachytherapy.
Summary: The goal of this randomized clinical trial is to find out whether giving an intravenous lidocaine + dexmedetomidine combination (LIDEX) during laparoscopic bariatric surgery can lower post-operative pain, inflammation, and oxidative stress in adults with obesity. The main questions it aims to answer are: * Pain control: Does LIDEX reduce pain 24 hours after surgery, as measured with the Internatio...
Summary: This is a Phase 1b, randomized, placebo/vehicle-controlled, double-blinded, multi-center trial. It is designed to assess the safety and efficacy of S. hominis A9 (ShA9) topical application as a treatment for atopic dermatitis (AD). The trial will enroll adults and adolescents with atopic dermatitis who are culture positive for S. aureus colonization. The primary safety objective of this study is t...
Related Latest Advances
Brand Information
- 120 g Tube – NDC 43538-900-12
(fluocinolone acetonide)
Cream, 0.025%
Not For Ophthalmic Use
PHARMACEUTICALS, INC.
