Urinary Incontinence Treatments
Find Urinary Incontinence Treatments
Brand Name
Darifenacin
View Brand InformationFDA approval date: September 01, 2016
Classification: Cholinergic Muscarinic Antagonist
Form: Tablet
What is Darifenacin?
Darifenacin extended-release tablets are muscarinic antagonist indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency. Darifenacin extended-release tablets are indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency.
Approved To Treat
Save this treatment for later
Not sure about your diagnosis?
Tired of the same old research?
Related Clinical Trials
A Randomized, Double-blind, Single Center, Phase 2 Study to Assess the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of 15 mg of Darifenacin Daily in Patients With Amyotrophic Lateral Sclerosis
Summary: Amyotrophic lateral sclerosis (ALS) is a progressive neurological disorder characterized by selective death of upper and lower motor neurons, which leads to severe disability and fatal outcomes. One of the major hallmarks of ALS is the denervation of neuromuscular junctions (NMJs), which is one of the earliest events seen in ALS patients and mouse models of ALS. Under healthy conditions, glial cel...
Related Latest Advances
Brand Information
Darifenacin (Darifenacin)
1INDICATIONS AND USAGE
Darifenacin extended-release tablets are indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency.
2DOSAGE AND ADMINISTRATION
The recommended starting dose of darifenacin extended-release tablets is 7.5 mg orally once daily. Based upon individual response, the dose may be increased to 15 mg once daily, as early as two weeks after starting therapy.
Darifenacin extended-release tablets should be taken orally once daily with water. Darifenacin extended-release tablets may be taken with or without food, and should be swallowed whole and not chewed, divided or crushed.
For patients with moderate hepatic impairment (Child-Pugh B) or when co-administered with potent CYP3A4 inhibitors (for example, ketoconazole, itraconazole, ritonavir, nelfinavir, clarithromycin and nefazadone), the daily dose of darifenacin extended-release tablets should not exceed 7.5 mg. Darifenacin extended-release tablets are not recommended for use in patients with severe hepatic impairment (Child-Pugh C)
3DOSAGE FORMS AND STRENGTHS
Darifenacin extended-release tablets 7.5 mg are white to off-white colored, round, biconvex, bevel edged, film coated tablets, debossed "202" on one side and plain on other side.
Darifenacin extended-release tablets 15 mg are light peach colored, round, biconvex, bevel edged, film coated tablets, debossed "203" on one side and plain on other side.
4CONTRAINDICATIONS
Darifenacin extended-release tablets are contraindicated in patients with, or at risk for, the following conditions:
- urinary retention
- gastric retention, or
- uncontrolled narrow-angle glaucoma.
5OVERDOSAGE
Overdosage with antimuscarinic agents, including darifenacin extended-release tablets, can result in severe antimuscarinic effects. Treatment should be symptomatic and supportive. In the event of overdosage, ECG monitoring is recommended. Darifenacin extended-release tablets has been administered in clinical trials at doses up to 75 mg (five times the maximum therapeutic dose) and signs of overdose were limited to abnormal vision.
6DESCRIPTION
Darifenacin extended-release tablet is an extended-release tablet for oral administration which contains 7.5 mg or 15 mg darifenacin as its hydrobromide salt. The active moiety, darifenacin, is a potent muscarinic receptor antagonist.
Chemically, darifenacin hydrobromide is
The structural formula is:
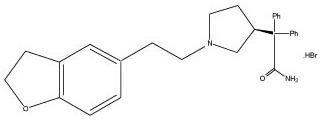
Darifenacin hydrobromide is a white to almost white to off-white powder, with a molecular weight of 507.5.
Darifenacin extended-release tablet is a once-a-day extended-release tablet and contains the following inactive ingredients: colloidal silicon dioxide, hypromellose (E15 LV), hypromellose (methocel K4M CR), magnesium stearate, microcrystalline cellulose, polyethylene glycol 400, talc and titanium dioxide. The 15 mg tablet also contains ferric oxide red and ferric oxide yellow.
7CLINICAL STUDIES
Darifenacin extended-release tablets were evaluated for the treatment of patients with overactive bladder with symptoms of urgency, urge urinary incontinence, and increased urinary frequency in three randomized, fixed-dose, placebo-controlled, multicenter, double-blind, 12-week studies (Studies 1, 2 and 3) and one randomized, double-blind, placebo-controlled, multicenter, dose-titration study (Study 4). For study eligibility in all four studies, patients with symptoms of overactive bladder for at least six months were required to demonstrate at least eight micturitions and at least one episode of urinary urgency per day, and at least five episodes of urge urinary incontinence per week. The majority of patients were white (94%) and female (84%), with a mean age of 58 years, range 19 to 93 years. Thirty-three percent of patients were greater than or equal to 65 years of age
Table 4 shows the efficacy data collected from 7- or 14-day voiding diaries in the three fixed-dose placebo-controlled studies of 1,059 patients treated with placebo, 7.5 mg or 15 mg once daily darifenacin for 12 weeks. A significant decrease in the primary endpoint, change from baseline in average weekly urge urinary incontinence episodes was observed in all three studies. Data is also shown for two secondary endpoints, change from baseline in the average number of micturitions per day (urinary frequency) and change from baseline in the average volume voided per micturition.
Table 5 shows the efficacy data from the dose-titration study in 395 patients who initially received 7.5 mg darifenacin or placebo daily with the option to increase to 15 mg darifenacin or placebo daily after two weeks.
As seen in Figures 2a, 2b and 2c, reductions in the number of urge incontinence episodes per week were observed within the first two weeks in patients treated with darifenacin 7.5 mg and 15 mg once daily compared to placebo. Further, these effects were sustained throughout the 12-week treatment period.
Figures 2a, 2b, 2c. Median Change from Baseline at Weeks 2, 6, 12 for Number of Urge Incontinence Episodes per Week (Studies 1, 2 and 3)

Figure 2a, Study 1

Figure 2b, Study 2

Figure 2c, Study 3
8HOW SUPPLIED/STORAGE AND HANDLING
Darifenacin extended-release tablets, 7.5 mg are white to off-white colored, round, biconvex, bevel edged, film coated tablets, debossed "202" on one side and plain on other side.
Bottle of 30.............................................................................................NDC 13668-202-30
Bottle of 90.............................................................................................NDC 13668-202-90
Bottle of 500..........................................................................................NDC 13668-202-05
Darifenacin extended-release tablets, 15 mg are light peach colored, round, biconvex, bevel edged, film coated tablets, debossed "203" on one side and plain on other side.
Bottle of 30........................................................................................... NDC 13668-203-30
Bottle of 90........................................................................................... NDC 13668-203-90
Bottle of 500..........................................................................................NDC 13668-203-05
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Protect from light.
Keep this and all drugs out of the reach of children.
9PATIENT COUNSELING INFORMATION
8100851See FDA-approved patient labeling (Patient Information).
Patients should be informed that anticholinergic agents, such as darifenacin extended-release tablets, may produce clinically significant adverse effects related to anticholinergic pharmacological activity including constipation, urinary retention and blurred vision. Heat prostration (due to decreased sweating) can occur when anticholinergics such as darifenacin extended-release tablets are used in a hot environment. Because anticholinergics, such as darifenacin extended-release tablets, may produce dizziness or blurred vision, patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until the drug's effects have been determined. Patients should read the patient information leaflet before starting therapy with darifenacin extended-release tablets.
Patients should be informed that darifenacin may produce clinically significant angioedema that may result in airway obstruction. Patients should be advised to promptly discontinue darifenacin therapy and seek immediate medical attention if they experience edema of the tongue or laryngopharynx, or difficulty breathing.
Darifenacin extended-release tablets should be taken once daily with water. They may be taken with or without food, and should be swallowed whole and not chewed, divided or crushed.

Manufactured by:
TORRENT PHARMACEUTICALS LTD., INDIA.
Manufactured for:
TORRENT PHARMA INC., Basking Ridge, NJ 07920
8106506 Revised: January 2026
10PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Darifenacin Extended-Release Tablets 7.5 mg: Bottle of 30 Tablets

Darifenacin Extended-Release Tablets 15 mg: Bottle of 30 Tablets

