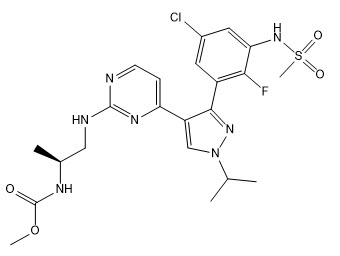
Braftovi
What is Braftovi (Encorafenib)?
Related Clinical Trials
Summary: This phase I trial tests the safety, best dose, and effectiveness of ZEN003694 in combination with cetuximab and encorafenib in treating patients with colorectal cancer that has not responded to previous treatment (refractory), that has come back after a period of improvement (relapsed), and that has spread from where it first started (primary site) to other places in the body (metastatic). ZEN003...
Summary: The purpose of this clinical trial (called the FLOTILLA study) is to give continued access to the study medicines, as well as safety follow-up, for participants in prior clinical trials of encorafenib and/or binimetinib. All participants who took part in earlier encorafenib and/or binimetinib studies may participate the FLOTILLA study if they are still benefiting from the use of the study medicine...
Summary: This research study is comparing two standard of care treatment options based on blood test results for participants who have metastatic colon cancer. The names of the potential treatments involved in this study are: * Active surveillance * FOLFIRI treatment * Nivolumab treatment * Encorafenib/Binimetinib/Cetuximab treatment * Trastuzumab + Pertuzumab
Related Latest Advances
Brand Information
- New Primary Malignancies
- Tumor Promotion in BRAF Wild-Type Tumors
- Cardiomyopathy
- Hepatotoxicity
- Hemorrhage
- Uveitis
- QT Prolongation
- Embryo-Fetal Toxicity
- Risks Associated with BRAFTOVI as a Single Agent
- Risks Associated with Combination Treatment