Brand Name
Juxtapid
Generic Name
Lomitapide
View Brand Information FDA approval date: January 03, 2013
Classification: Microsomal Triglyceride Transfer Protein Inhibitor
Form: Capsule
What is Juxtapid (Lomitapide)?
JUXTAPID is a microsomal triglyceride transfer protein inhibitor indicated as an adjunct to a low-fat diet and other lipid-lowering treatments, including LDL apheresis where available, to reduce low-density lipoprotein cholesterol , total cholesterol , apolipoprotein B , and non-high-density lipoprotein cholesterol in patients with homozygous familial hypercholesterolemia . Limitations of Use The safety and effectiveness of JUXTAPID have not been established in patients with hypercholesterolemia who do not have HoFH, including those with heterozygous familial hypercholesterolemia . The effect of JUXTAPID on cardiovascular morbidity and mortality has not been determined .
Approved To Treat
Top Global Experts
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
Evaluation of the Effect of Lomitapide Treatment on Major Adverse Cardiovascular Events (MACE) in Patients With Homozygous Familial Hypercholesterolemia: A Multicenter, Retrospective and Prospective Observational Study
Summary: This observational, multicenter, retrospective and prospective study aims to evaluate the effect of lomitapide treatment on Major Adverse Cardiovascular Events (MACE) in patients with Homozygous Familial Hypercholesterolemia (HoFH). HoFH is a rare genetic disorder characterized by extremely high levels of LDL cholesterol (LDL-C), leading to an increased risk of early cardiovascular diseases. Lomit...
LOWER: Lomitapide Observational Worldwide Evaluation Registry
Summary: This global product exposure registry is a multicentre, long-term, prospective, observational cohort study (exposure registry), designed to evaluate the long term safety and effectiveness of lomitapide.
Related Latest Advances
Brand Information
Juxtapid (lomitapide mesylate)
WARNING: RISK OF HEPATOTOXICITY
JUXTAPID can cause elevations in transaminases. In the adult clinical trial, 10 (34%) of the 29 patients treated with JUXTAPID had at least one elevation in alanine aminotransferase (ALT) or aspartate aminotransferase (AST) ≥3 times the upper limit of normal (ULN). There were no concomitant clinically meaningful elevations of total bilirubin, international normalized ratio (INR), or alkaline phosphatase. In the pediatric clinical trial (5 to 17 years of age), 6 (14%) of the 43 patients experienced elevations in ALT and/or AST ≥ 3 times ULN. No concomitant clinically meaningful elevations in total bilirubin or alkaline phosphatase were observed
JUXTAPID also increases hepatic fat, with or without concomitant increases in transaminases. The median absolute increase in hepatic fat in adult patients was 6% after both 26 and 78 weeks of treatment, from 1% at baseline, measured by magnetic resonance spectroscopy (MRS). The median absolute increase in hepatic fat in pediatric patients aged 5 to 17 years was 4% after 24 weeks and 104 weeks of treatment, from 3% at baseline, measured by nuclear magnetic resonance (NMR). Hepatic steatosis associated with JUXTAPID treatment may be a risk factor for progressive liver disease, including steatohepatitis and cirrhosis
Measure ALT, AST, alkaline phosphatase, and total bilirubin before initiating treatment and then ALT and AST regularly as recommended. During treatment, adjust the dose of JUXTAPID if the ALT or AST are ≥3 times ULN. Discontinue JUXTAPID for clinically significant liver toxicity [
Because of the risk of hepatotoxicity, JUXTAPID is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the JUXTAPID REMS Program
1INDICATIONS AND USAGE
JUXTAPID is indicated as an adjunct to a low-fat diet and exercise and other low density lipoprotein cholesterol (LDL-C) therapies to reduce LDL-C in adults and pediatric patients aged 2 years and older with homozygous familial hypercholesterolemia (HoFH).
2DOSAGE FORMS AND STRENGTHS
Capsules:
- 2 mg: Grey/grey hard gelatin capsule printed with black ink "A733" and "2 mg"
- 5 mg: Orange/orange hard gelatin capsule printed with black ink "A733" and "5 mg"
- 10 mg: Orange/white hard gelatin capsule printed with black ink "A733" and "10 mg"
- 20 mg: White/white hard gelatin capsule printed with black ink "A733" and "20 mg"
- 30 mg: Orange/yellow hard gelatin capsule printed with black ink "A733" and "30 mg"
3CONTRAINDICATIONS
JUXTAPID is contraindicated in the following conditions:
- Pregnancy
- Concomitant administration of JUXTAPID with moderate or strong CYP3A4 inhibitors, as this can increase JUXTAPID exposure
- Patients with moderate or severe hepatic impairment (based on Child-Pugh category B or C) and patients with active liver disease, including unexplained persistent elevations of serum transaminases
4ADVERSE REACTIONS
The following important adverse reactions have been observed and are discussed in detail in other sections of the label:
- Risk of hepatotoxicity
- Reduced absorption of fat-soluble vitamins, and serum fatty acids
- Gastrointestinal adverse reactions
4.1Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults with HoFH
One single-arm, open-label, 78-week trial has been conducted in 29 adult patients with HoFH, 23 of whom completed at least one year of treatment. The initial dosage of JUXTAPID was 5 mg daily, with titration up to 60 mg daily during an 18-week period based on safety and tolerability. In this trial, the mean age was 31 years (range, 18 to 55 years), 16 (55%) patients were male, 25 (86%) patients were White, 2 (7%) were Asian, 1 (3%) was Black or African American, and 1 (3%) was multi-racial
Five (17%) of the 29 patients discontinued treatment due to an adverse reaction. The adverse reactions that contributed to treatment discontinuations included diarrhea (2 patients; 7%) and abdominal pain, nausea, gastroenteritis, weight loss, headache, and difficulty controlling INR on warfarin (1 patient each; 3%).
The most common adverse reactions were gastrointestinal, reported by 27 (93%) of 29 patients. Adverse reactions reported by ≥8 (28%) patients in the clinical trial included diarrhea, nausea, vomiting, dyspepsia, and abdominal pain. Other common adverse reactions, reported by 5 to 7 (17 to 24%) patients, included weight loss, abdominal discomfort, abdominal distension, constipation, flatulence, increased ALT, chest pain, influenza, nasopharyngitis, and fatigue.
The adverse reactions reported in at least 10% of adult patients are presented in Table 8.
Adverse reactions of severe intensity were reported by 8 (28%) of 29 patients, with the most common being diarrhea (4 patients, 14%), vomiting (3 patients, 10%), increased ALT or hepatotoxicity (3 patients, 10%), and abdominal pain, distension, and/or discomfort (2 patients, 7%).
Pediatric Patients with HoFH Aged 5 to 17 years
A single-arm, open label, multinational, 104-week trial was conducted in 43 pediatric patients with HoFH aged 5 to 17 years. Thirty-nine of the patients completed the trial. The dose of JUXTAPID was escalated from an age-dependent starting dose to a maximum tolerated dose (MTD) as applicable to the pediatric age group and based on acceptable safety and tolerability criteria, in addition to LDL-C goals
Transaminase Elevations
During the adult clinical trial, 10 (34%) of 29 patients had at least one elevation in ALT and/or AST ≥3 times ULN (see
Among the 19 adult patients who enrolled in an extension trial following the adult clinical trial, one discontinued because of increased transaminases that persisted despite several dose reductions, and one temporarily discontinued because of markedly elevated transaminases (ALT 24 times ULN, AST 13 times ULN) that had several possible causes, including a drug-drug interaction between JUXTAPID and the strong CYP3A4 inhibitor clarithromycin
In the pediatric trial, 6 patients experienced elevations in ALT and/or AST ≥3 times ULN (see
Hepatic Steatosis
Hepatic fat was prospectively measured using magnetic resonance spectroscopy (MRS) in all eligible patients during the adult clinical trial. After 26 weeks, the median absolute increase in hepatic fat from baseline was 6%, and the mean absolute increase was 8% (range, 0% to 30%). After 78 weeks, the median absolute increase in hepatic fat from baseline was 6%, and the mean absolute increase was 7% (range, 0% to 18%). Among the 23 patients with evaluable data on at least one occasion during the trial, 18 (78%) exhibited an increase in hepatic fat >5% and 3 (13%) exhibited an increase >20%. Data from individuals who had repeat measurements after stopping JUXTAPID show that hepatic fat accumulation is reversible, but whether histological sequelae remain is unknown.
In the pediatric clinical trial, the median absolute increase in hepatic fat was 4% after 24 weeks and 104 weeks, from 3% at baseline, measured by NMR. Among the 19 patients with hepatic fat measured by NMR on at least one occasion during the trial, 8 (42%) patients exhibited an increase in hepatic fat to >10% including 1 (5%) patient with an increase to >20%. Data from pediatric patients with follow-up measurements after Week 104 suggest that hepatic fat accumulation is reversable after stopping treatment with JUXTAPID, but whether histological sequelae remain is unknown, especially after long term use.
4.2Postmarketing Experience
The following adverse reactions have been identified during post-approval use of JUXTAPID. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to JUXTAPID exposure.
Musculoskeletal: Myalgia
Skin reactions: Alopecia
5OVERDOSAGE
There is no specific treatment in the event of overdose of JUXTAPID. In the event of overdose, the patient should be treated symptomatically, and supportive measures instituted as required. Liver-related tests should be monitored. Hemodialysis is unlikely to be beneficial given that lomitapide is highly protein-bound.
6DESCRIPTION
JUXTAPID capsules contain lomitapide mesylate, a synthetic lipid-lowering agent for oral administration.
The chemical name of lomitapide mesylate is N-(2,2,2-trifluoroethyl)-9-[4-[4-[[[4'a-(trifluoromethyl)[1,1'-biphenyl]-2-yl]carbonyl]amino]-1-piperidinyl]butyl]-9

The empirical formula for lomitapide mesylate is C
Lomitapide mesylate is a white to off-white powder that is slightly soluble in aqueous solutions of pH 2 to 5. Lomitapide mesylate is freely soluble in acetone, ethanol, and methanol; soluble in 2-butanol, methylene chloride, and acetonitrile; sparingly soluble in 1-octanol and 2-propanol; slightly soluble in ethyl acetate; and insoluble in heptane.
Each JUXTAPID capsule contains lomitapide mesylate equivalent to 2, 5, 10, 20, or 30 mg lomitapide free base and the following inactive ingredients: pregelatinized starch, sodium starch glycolate, microcrystalline cellulose, lactose monohydrate, silicon dioxide and magnesium stearate. The capsule shells of all strengths contain gelatin and titanium dioxide; the 2 mg capsules contain black iron oxide; the 5 mg, 10 mg and 30 mg capsules also contain red iron oxide; and the 30 mg capsules also contain yellow iron oxide. The imprinting ink contains shellac, black iron oxide, and propylene glycol.
7CLINICAL STUDIES
Adults with HoFH
The safety and effectiveness of JUXTAPID as an adjunct to a low-fat diet and other lipid-lowering treatments, including LDL apheresis where available, were evaluated in a multinational, single-arm, open-label, 78-week trial involving 29 adults with HoFH. A diagnosis of HoFH was defined by the presence of at least one of the following clinical criteria: (1) documented functional mutation(s) in both LDL receptor alleles or alleles known to affect LDL receptor functionality, or (2) skin fibroblast LDL receptor activity <20% normal, or (3) untreated TC >500 mg/dL and TG <300 mg/dL
Among the 29 patients enrolled, the mean age was 31 years (range, 18 to 55 years), 16 (55%) were male, and the majority (86%) were White. The mean body mass index (BMI) was 25.8 kg/m
After a six-week run-in period to stabilize lipid-lowering treatments, including the establishment of an LDL apheresis schedule if applicable, JUXTAPID was initiated at 5 mg daily and titrated to daily doses of 10 mg, 20 mg, 40 mg, and 60 mg at weeks 2, 6, 10, and 14, respectively, based on tolerability and acceptable levels of transaminases. Patients were instructed to maintain a low-fat diet (<20% calories from fat) and to take dietary supplements that provided approximately 400 international units vitamin E, 210 mg ALA, 200 mg linoleic acid, 110 mg EPA, and 80 mg (DHA per day
Twenty-three (79%) patients completed the efficacy endpoint at Week 26, all of whom went on to complete 78 weeks of treatment. Adverse events contributed to premature discontinuation for five patients
The primary efficacy endpoint was percent change in LDL-C from baseline to Week 26. At Week 26, the mean and median percent changes in LDL-C from baseline were -40% (paired t-test p<0.001) and -50%, respectively, based on the intent-to-treat population with last observation carried forward (LOCF) for patients who discontinued prematurely. The mean percent change in LDL-C from baseline through Week 26 is shown in Figure 1 for the 23 patients who completed the efficacy period.
Changes in lipids and lipoproteins through the efficacy endpoint at Week 26 are presented in Table 14.
After Week 26, during the safety phase of the trial, adjustments to concomitant lipid-lowering treatments were allowed. For the study population overall, average reductions in LDL-C, TC, apo B, and non-HDL-C were sustained during chronic therapy.
Pediatric Patients with HoFH
A single-arm, open-label, 104-week trial evaluated the efficacy of JUXTAPID when co-administered with a low-fat diet in pediatric patients aged 5 to 17 years of age with HoFH on stable lipid lowering therapy (LLT), including low density lipoprotein (LDL) apheresis, when applicable. A diagnosis of HoFH was defined by any of the following criteria: (1) genetic confirmation of 2 mutant alleles at the
A total of 43 patients (19 males, 44% and 24 females, 56%) between 5 and 17 years of age (mean age of 11 years) were treated in this trial. The majority (98%) of patients were White (one [2%] patient was Black or African American and one [2%] patient identified as Hispanic/Latino ethnicity). The mean BMI was 19 kg/m
The trial consisted of a 6-week run-in period, followed by a 24-week efficacy phase, and then an 80-week safety phase. The dose of JUXTAPID was escalated from an age-dependent starting dose to a maximum tolerated dose as applicable to the pediatric age group (see
Forty-six (46) patients were enrolled, of whom 43 completed the run-in period, 41 completed Week 24, and 39 completed the safety phase. In the efficacy phase, 95% of patients aged 5 to 10 years received the MTD of 20 mg, 76% of patients aged 11 to 15 years received the MTD of 40 mg, and 50% of patients aged 16 to 17 years received the MTD of 60 mg but had to down-titrate soon after reaching this dose.
The primary endpoint was percent change in LDL-C from baseline to Week 24. At Week 24, the mean percent change in LDL-C from baseline was -49% (95% CI: -59%, -38%) (see
Figure 2: Observed Mean Percent Change in LDL-C from Baseline in Pediatric Patients Aged 5 to 17 years

Subgroup analysis was carried out on patients aged 5 to 10 years (N=20) and 11 to 17 years (N=23). Mean decreases from baseline in LDL-C at Week 24 were 52% and 46%, respectively. Subgroup analysis also demonstrated mean decreases from baseline in LDL-C at Week 24 of approximately 36% in patients receiving LDL apheresis at baseline and 62% in patients not receiving LDL apheresis at baseline. During the long-term treatment (over 2 years), mean LDL-C levels in the LDL apheresis group showed a gradual increase over time.
The key secondary endpoint was the mean percent change in lipid parameters (non-HDL-C, total cholesterol, VLDL-C, apo B, and triglycerides) from baseline to Week 24. At Week 24, there were mean percent decreases from baseline in each of the lipid parameters assessed (summarized in Table 15).
Average reductions in LDL-C, TC, apo B, TG, non-HDL-C, and VLDL-C were sustained during long-term treatment (over 2 years) particularly in patients not receiving LDL apheresis at baseline.
8HOW SUPPLIED / STORAGE AND HANDLING
How Supplied
JUXTAPID strengths are available as follows:
9PATIENT COUNSELING INFORMATION
See FDA-approved labeling (
Patients or their caregiver(s) should be informed that a registry for patients taking JUXTAPID has been established to monitor and evaluate the long-term effects of JUXTAPID. Patients or their caregiver(s) are encouraged to participate in the registry and should be informed that their participation is voluntary. For information regarding the registry program visit www.JUXTAPID.com or call 1-877-902-4099.
Advise patients of the following:
Risk of Hepatotoxicity
- JUXTAPID can cause both elevations in transaminases and hepatic steatosis. Discuss with patients or their caregiver(s) about the importance of monitoring of liver-related tests before taking JUXTAPID, prior to each dose escalation, and periodically thereafter.
- Advise patients of the potential for increased risk of liver injury if alcohol is consumed while taking JUXTAPID. It is recommended that patients taking JUXTAPID limit consumption to not more than one alcoholic drink per day.
- JUXTAPID is commonly associated with nausea, vomiting, and abdominal pain. Advise patients or their caregiver(s) to promptly report these symptoms if they increase in severity, persist, or change in the character, as they might reflect liver injury. Patients or their caregiver(s) should also report any other symptoms of possible liver injury, including fever, jaundice, lethargy, or flu-like symptoms.
JUXTAPID REMS Program
JUXTAPID is only available through a restricted program called JUXTAPID REMS Program and therefore, JUXTAPID is only available from certified pharmacies that are enrolled in the program.
Embryofetal Toxicity
- JUXTAPID is contraindicated in pregnancy since it may cause fetal harm. Advise female patients who become pregnant to discontinue JUXTAPID and inform their healthcare provider of a known or suspected pregnancy.
- Advise female patients of reproductive potential to use effective contraception during treatment with JUXTAPID and for two weeks after the final dose.
- Advise patients who are taking oral contraceptives and experience vomiting or diarrhea while taking JUXTAPID to use an effective alternative contraceptive method until 7 days after resolution of symptoms
Lactation
Advise female patients not to breastfeed during treatment with JUXTAPID
Dietary Supplements
- Inform patients or their caregiver(s) that JUXTAPID may reduce the absorption of fat-soluble nutrients. Instruct patients or their caregiver(s) to take daily nutritional supplements that contain the dosages of vitamin E and essential fatty acids recommended in the
Gastrointestinal Adverse Reactions
- Inform patients or their caregiver(s) that gastrointestinal adverse reactions are common with JUXTAPID. These include, but are not limited to, diarrhea, nausea/vomiting, abdominal pain/discomfort, flatulence, and constipation. Advise patients or their caregiver(s) to adhere to low-fat diet supplying <20% of energy from fat or less than 30 grams of fat, whichever is less, to reduce the risk of gastrointestinal adverse reactions.
- Instruct patients or their caregiver(s) to stop JUXTAPID and contact their healthcare provider if severe diarrhea occurs or if they experience symptoms of volume depletion such as lightheadedness, decreased urine output, or tiredness.
- Inform patients or their caregiver(s) that taking JUXTAPID with food may adversely impact gastrointestinal tolerability; therefore, they should take JUXTAPID at least 2 hours after the evening meal, swallowing each capsule whole, with water. Inform patients or their caregiver(s) that if the patient is unable to swallow the intact capsule(s), the capsule(s) can be opened and the contents sprinkled on 1 tablespoon of apple sauce or mashed banana, which are fat free. For younger patients who require a smaller spoon for administration, inform the patient or their caregiver(s) to sprinkle the capsule content onto 1 tablespoon of apple sauce or mashed banana and administer the entire amount using the smaller spoon.
- Absorption of oral medications may be affected in patients who develop diarrhea or vomiting. For example, hormone absorption from oral contraceptives may be incomplete, warranting the use of additional contraceptive methods. Patients who develop these symptoms should seek advice from their healthcare provider.
Drug Interactions
- Instruct patients or their caregiver(s) to avoid food or drinks containing grapefruit during JUXTAPID treatment.
- Because multiple drug-drug interactions have been described with JUXTAPID, advise patients or their caregiver(s) to tell their healthcare provider(s) about all medications, nutritional supplements, and vitamins that they are taking or may be taking while taking JUXTAPID.
- Advise patients taking simvastatin or lovastatin that JUXTAPID may cause myopathy. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever.
Missed Doses
- If a dose of JUXTAPID is missed, the normal dose should be taken at the usual time the next day. If dosing is interrupted for more than a week, tell patients or their caregiver(s) to contact their healthcare provider before restarting treatment.
10PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Label
NDC 10122-402-28
28 capsules
Juxtapid
2 mg

11PRINCIPAL DISPLAY PANEL - 2 mg Capsule Bottle Carton
NDC 10122-402-28
28 capsules
Juxtapid
2 mg
Attention Pharmacist:

12PRINCIPAL DISPLAY PANEL - 5 mg Capsule Bottle Label
NDC 10122-405-28
28 capsules
Juxtapid
5 mg
Chiesi

13PRINCIPAL DISPLAY PANEL - 5 mg Capsule Bottle Carton
NDC 10122-405-28
28 capsules
Juxtapid
5 mg
Attention Pharmacist:
Chiesi

14PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label
NDC 10122-410-28
28 capsules
Juxtapid
10 mg
Chiesi

15PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Carton
NDC 10122-410-28
28 capsules
Juxtapid
10 mg
Attention Pharmacist:
Chiesi

16PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label
NDC 10122-420-28
28 capsules
Juxtapid
20 mg
Chiesi
17PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Carton
NDC 10122-420-28
28 capsules
Juxtapid
20 mg
Attention Pharmacist:
Chiesi

18PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label
NDC 10122-430-28
28 capsules
Juxtapid
30 mg
Chiesi

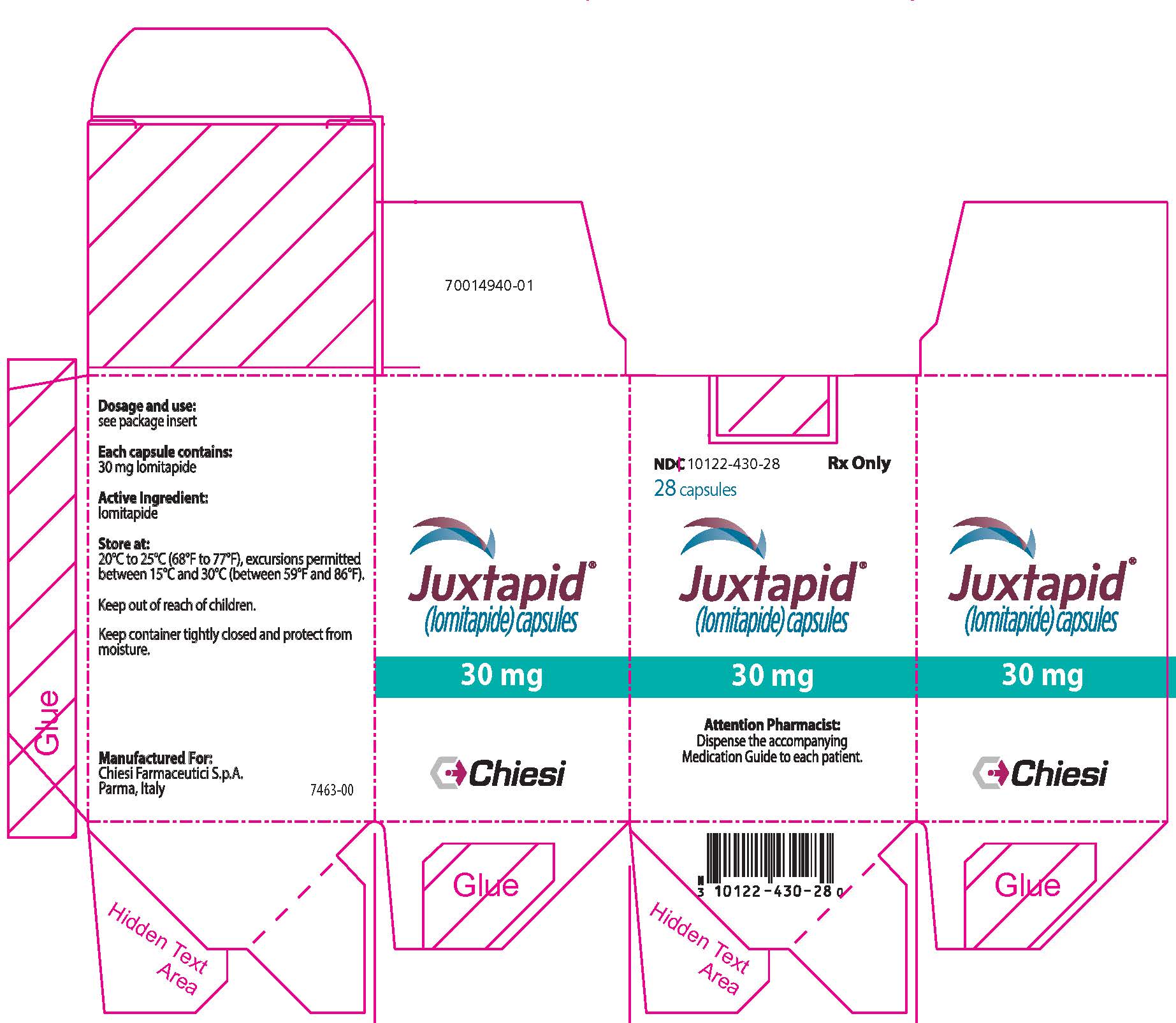
19PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Carton
NDC 10122-430-28
28 capsules
Juxtapid
30 mg
Attention Pharmacist:
Chiesi
