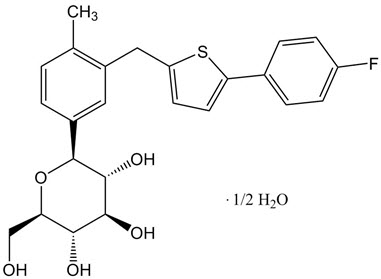
Invokana
What is Invokana (Canagliflozin)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: Acute kidney disease (AKD) happens between 7 and 90 days after an initial kidney injury (AKI). This period is crucial because it can determine whether the condition worsens into chronic kidney disease (CKD). Despite knowing this, there is no proven treatment to improve outcomes for people with AKD. Recent studies have shown that drugs called sodium-glucose cotransporter 2 (SGLT2) inhibitors can sl...
Summary: This is a phase II, proof of concept, placebo-controlled, randomized clinical trial, assessing the effect of canagliflozin on cardiac structure and function in patients with advanced renal disease, including those on maintenance dialysis. Our primary aim is to determine the effect of canagliflozin on cardiac structure and function in patients with advanced chronic kidney disease (CKD), compared wi...
Summary: PRECIDENTD is a randomized, open label, pragmatic clinical trial designed to compare rates of the total number of cardiovascular, kidney, and death events among two alternative treatments for patients with type 2 diabetes (T2D) and either established atherosclerotic cardiovascular disease (ASCVD) or at high risk for ASCVD. To accomplish this objective, we will randomly assign 6,000 patients with e...
Related Latest Advances
Brand Information
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease (CVD).
- to reduce the risk of end-stage kidney disease (ESKD), doubling of serum creatinine, cardiovascular (CV) death, and hospitalization for heart failure in adults with type 2 diabetes mellitus and diabetic nephropathy with albuminuria greater than 300 mg/day.
- INVOKANA 100 mg tablets are yellow, capsule-shaped, tablets with "CFZ" on one side and "100" on the other side.
- INVOKANA 300 mg tablets are white, capsule-shaped, tablets with "CFZ" on one side and "300" on the other side.
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes and Other Ketoacidosis
- Lower Limb Amputation
- Volume Depletion
- Urosepsis and Pyelonephritis
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
- Necrotizing Fasciitis of the Perineum (Fournier's gangrene)
- Genital Mycotic Infections
- Hypersensitivity Reactions
- Bone Fracture