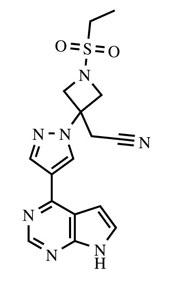
Olumiant
What is Olumiant (Baricitinib)?
Top Global Experts
Related Clinical Trials
Background: Autosomal dominant hyper-IgE syndrome (HIES), also called Job syndrome, is a genetic disorder that affects the immune system. It can cause skin and lung infections and problems with blood vessels, connective tissues, and bones. People with HIES often have lupus-like disease or atopic dermatitis (skin rash). Researchers want to know if a drug approved to treat other immune system diseases (bariciti...
Summary: The purpose of the present study is to study the effect of baricitinib administration on outcome of participants with moderate and severe traumatic intracerebral hemorrhage/contusions. A multi-center randomized control trial will be conducted. Participants with a radiological diagnosis of traumatic intracerebral hemorrhage/contusions and an initial GCS score of 5-12 will be screened and enrolled i...
Background: Kohlmeier-Degos (KD) is a rare disease that causes inflammation and blood clots, leading to blockages in small blood vessels. These blockages can result in K-D lesions throughout the body, affecting the skin, lungs, heart, spinal cord, and brain. KD can be fatal. No treatment exists for this disease.
Related Latest Advances
Brand Information
- 1 mg tablet contains a recessed area on each face of the tablet surface, is very light pink, round, debossed with “Lilly” on one side and “1” on the other.
- 2 mg tablet contains a recessed area on each face of the tablet surface, is light pink, oblong, debossed with “Lilly” on one side and “2” on the other.
- 4 mg tablet contains a recessed area on each face of the tablet surface, is medium pink, round, debossed with “Lilly” on one side and “4” on the other.
- Serious Infections
- Mortality
- Malignancy and Lymphoproliferative Disorders
- Major Adverse Cardiovascular Events
- Thrombosis
- Hypersensitivity
- Gastrointestinal Perforations
- Laboratory Abnormalities