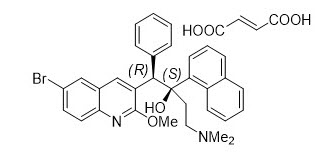
Sirturo
What is Sirturo (Bedaquiline)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: This study aims to assess quabodepistat-based treatment regimens for RR/MDR-TB. The study will enroll adults and adolescents with rifampicin-resistant or multidrug-resistant pulmonary TB. The main goal is to see if a new drug called quabodepistat, when combined with other TB drugs, can shorten treatment duration to 4 months and be as effective and safer than current WHO endorsed treatment regimen ...
Summary: A5409/RAD-TB is an adaptive Phase 2 randomized, controlled, open-label, dose-ranging, platform protocol to evaluate the safety and efficacy of multidrug regimens for the treatment of adults with drug-susceptible pulmonary tuberculosis (TB). A5409 hypothesizes that novel regimens for the treatment of pulmonary tuberculosis will result in superior early efficacy, as determined by longitudinal mycoba...
Summary: This is a multicenter, randomized, open-label, active-controlled clinical study designed to evaluate the efficacy, safety, and pharmacokinetic characteristics of different doses of JDB0131 benzenesulfonate tablets compared with delamanid in combination with bedaquiline, linezolid, levofloxacin (moxifloxacin)/clofazimine, etc. in the treatment of patients with drug-resistant (including rifampicin-r...
Related Latest Advances
Brand Information
- SIRTURO 20 mg tablet: uncoated, white to almost white oblong functionally scored tablet, with a score line on both sides, debossed with "2" and "0" on one side and plain on the other side.
- SIRTURO 100 mg tablet: uncoated, white to almost white round biconvex tablet with debossing of "T" over "207" on one side and "100" on the other side.
- QTc Prolongation
- Mortality Imbalance in Clinical Trials
- Hepatotoxicity
- Drug Interactions

(bedaquiline) tablets
188 Tablets

