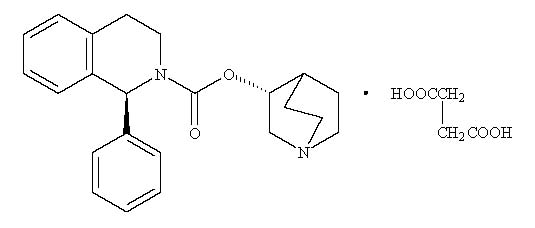
VESIcare
What is VESIcare (Solifenacin)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: The primary objective is to evaluate if (1) combination therapy of solifenacin and mirabegron in low doses is superior to monotherapy of solifenacin in high dose and if (2) combination therapy of mirabegron and solifenacin in low doses is superior to monotherapy of mirabegron in high dose in treatment of daytime urinary incontinence among children aged 5 to 14 years who are none complete responder...
Summary: The primary objective is to investigate if abrupt withdrawal versus gradual withdrawal of pharmacotherapy (solifenacin and/or mirabegron) influences the risk of recurrence of incontinence. Children aged 5-14 years diagnosed with urinary incontinence, treated with pharmacotherapy of solifenacin and/or mirabegron and ready for withdrawal will be randomized 1:1 to either abrupt or gradual withdrawal,...
Summary: The goal of this clinical trial is to compare combination of Solifenacin and MIrabegron in treatment of primary mono-symptomatic nocturnal enuresis in children. It will also learn about the safety of that combination. The main questions it aims to answer are: Does combination of Solifenacin and MIrabegron lower the number of wet nights? What medical problems do participants have when taking combin...
Related Latest Advances
Brand Information
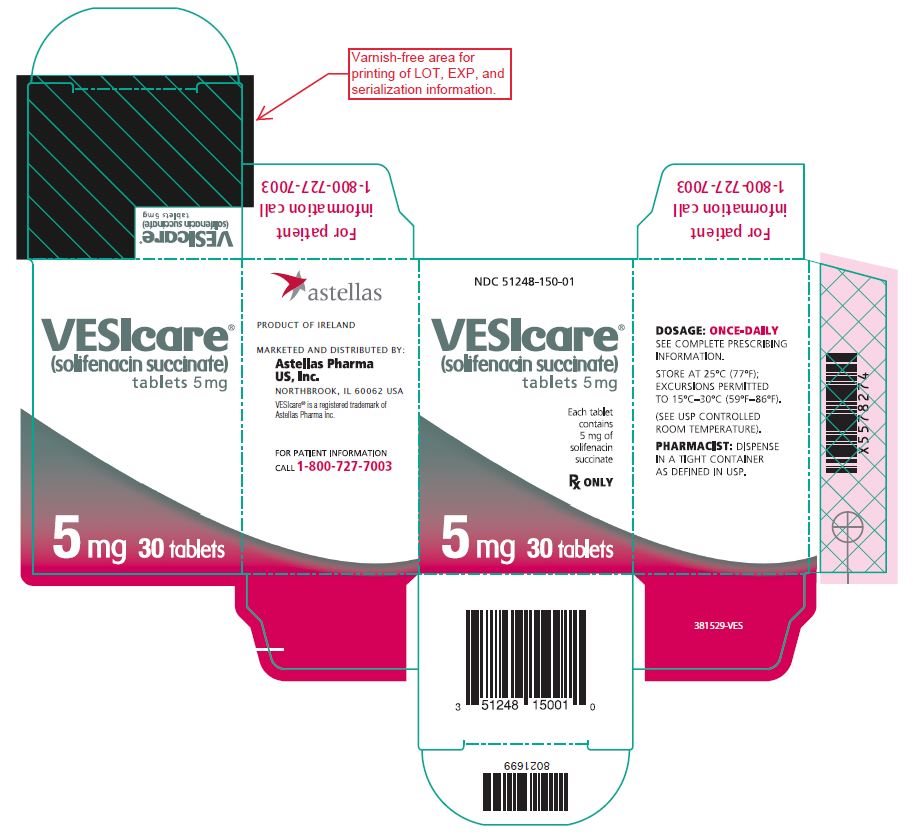
- 5 mg: round, light yellow, debossed with 150
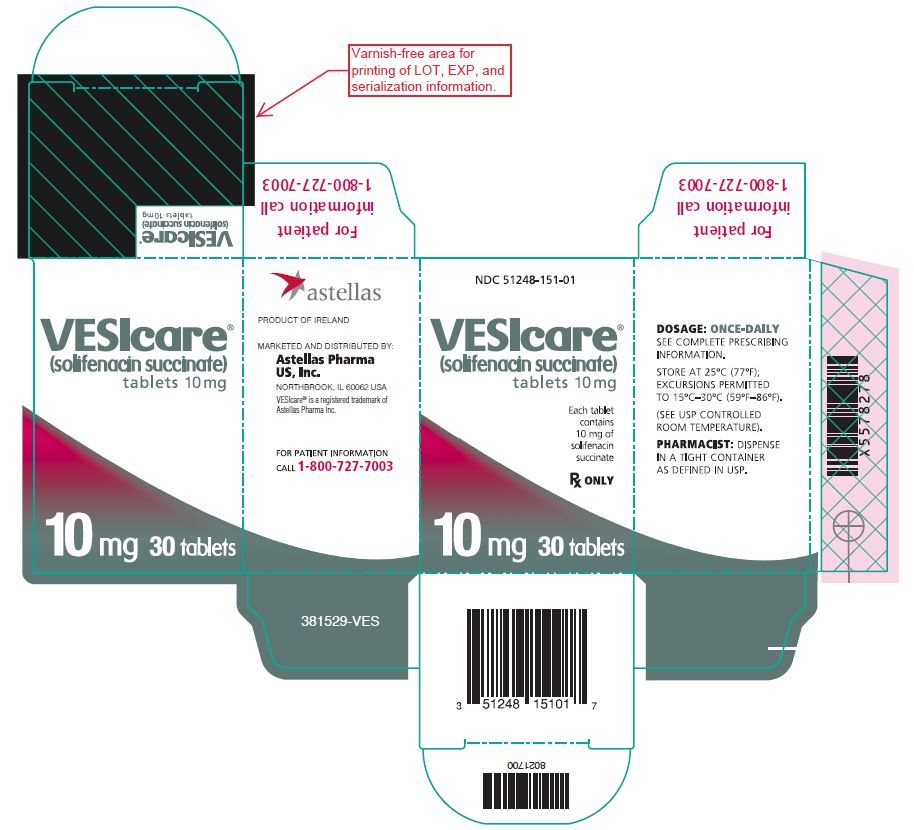
- 10 mg: round, light pink, debossed with 151
- With urinary retention
- With gastric retention
- With uncontrolled narrow-angle glaucoma
- Who have demonstrated hypersensitivity to solifenacin succinate or the inactive ingredients in VESIcare. Reported adverse reactions have included anaphylaxis and angioedema
- Bottle of 30 NDC 51248-150-01
- Bottle of 30 NDC 51248-151-01
- Bottle of 90 NDC 51248-151-03