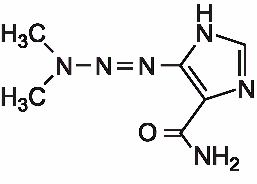
Dacarbazine
View Brand InformationWhat is Dacarbazine?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: The purpose of this study is to determine the ability of letrozole to penetrate the blood brain barrier and concentrate in gliomas.
Summary: This trial is a multi-center, non-randomized, open-label Phase I/II study evaluating the feasibility and efficacy of vincristine, irinotecan, temozolomide, and atezolizumab in children with relapsed/refractory solid tumors.
Summary: Investigators are testing new experimental drug combinations such as the combination of vorinostat, vincristine, irinotecan, and temozolomide in the hopes of finding a drug that may be effective against tumors that have come back or that have not responded to standard therapy. The goals of this study are: * To find the highest safe dose of vorinostat that can be given together with vincristine, ir...
Related Latest Advances
Brand Information