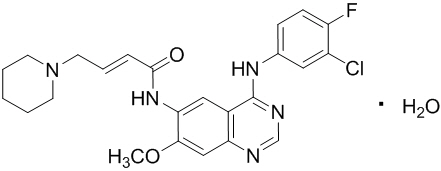
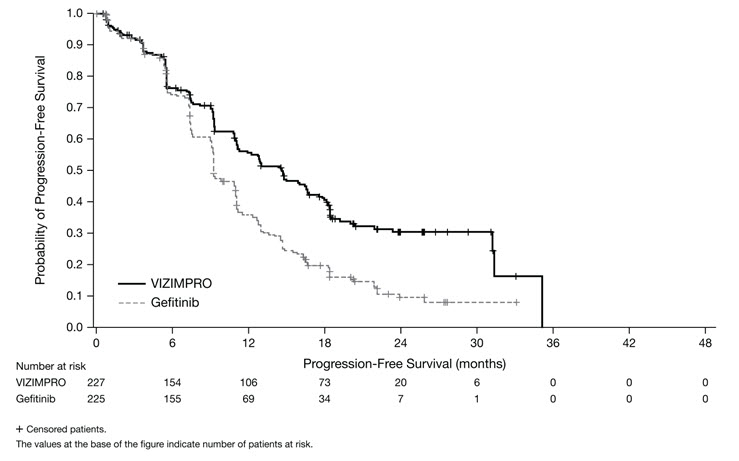
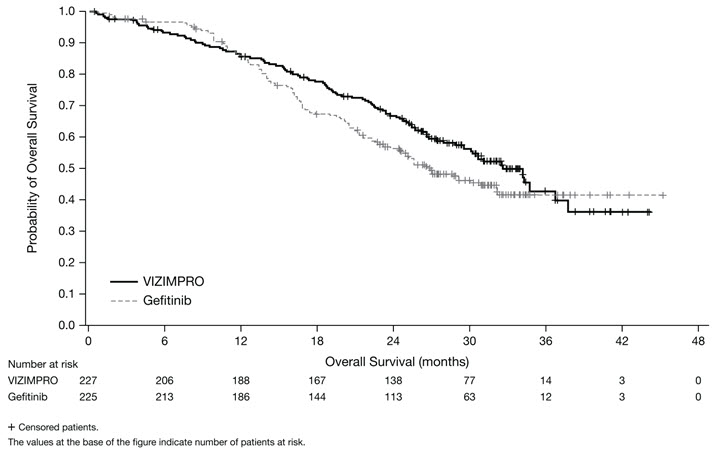
Vizimpro
What is Vizimpro (Dacomitinib)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: A Phase II, open label, non-randomized, multiple-arm, single-center clinical trial in patients with advanced rare solid tumors who failed to standard treatment.
Summary: The purpose of this study is to evaluate the optimal sequence of EGFR-inhibitors in lung cancer patients with EGFR-positive tumors not amenable for curative treatment. Life quality, adverse effects and tumor response will be evaluated and analyses of obtained blood and tumor samples will be performed to identify molecular profiles and biomarkers that can be used for treatment decisions.
Summary: This is a prospective, non-randomized clinical trial that aims to describe the efficacy and toxicity of commercially available, targeted anticancer drugs\* prescribed for treatment of patients with advanced cancer with a potentially actionable variant as revealed by a genomic or protein expression test. The study also aims to simplify patient access to approved targeted therapies that are contribu...
Related Latest Advances
Brand Information
- 45 mg: blue film-coated, immediate release, round biconvex tablet, debossed with "Pfizer" on one side and "DCB45" on the other side.
- 30 mg: blue film-coated, immediate release, round biconvex tablet, debossed with "Pfizer" on one side and "DCB30" on the other side.
- 15 mg: blue film-coated, immediate release, round biconvex tablet, debossed with "Pfizer" on one side and "DCB15" on the other side.
- Interstitial Lung Disease
- Diarrhea
- Dermatologic Adverse Reactions