ARIPiprazole
What is Abilify Asimtufii (ARIPiprazole)?
Top Global Experts
Related Clinical Trials
Summary: The effectiveness and efficacy of the combination of pharmacotherapy with the two new recovery-oriented programs, RECOVERYTRSGR for patients with treatment- resistant schizophrenia and RECOVERYTRSBDGR for patients with treatment- resistant bipolar disorder.
Summary: This project aims to investigate the effectiveness of existing common antidepressants and to provide new evidence for depressed children and adolescents who are not responding to their first treatment.
Summary: This randomized, single-blind (assessor-blind) controlled trial aims to investigate the efficacy of aripiprazole as an augmentation strategy for treating pathological rumination in patients with major depressive disorder (MDD). Pathological rumination-defined as repetitive, intrusive, and uncontrollable negative thinking-has been identified as a major transdiagnostic risk factor for the developmen...
Related Latest Advances
Brand Information
- for the treatment of schizophrenia in adults
- for maintenance monotherapy treatment of bipolar I disorder in adults
- Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use
- Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis
- Neuroleptic Malignant Syndrome
- Tardive Dyskinesia
- Metabolic Changes
- Pathological Gambling and Other Compulsive Behaviors
- Orthostatic Hypotension and Syncope
- Falls
- Leukopenia, Neutropenia, and Agranulocytosis
- Seizures
- Potential for Cognitive and Motor Impairment
- Body Temperature Regulation
- Dysphagia
- Read the complete instructions for preparation and administration below before administering ABILIFY ASIMTUFII.
- To be prepared and administered only by a healthcare professional once every two months
- ABILIFY ASIMTUFII pre-filled syringe is
- For gluteal intramuscular injection only. Do not administer by any other route.
- Prior to administration, visually inspect ABILIFY ASIMTUFII pre-filled syringe for particulate matter and discoloration. The suspension should appear to be a uniform, homogeneous suspension that is opaque and milky-white in color. Do not use ABILIFY ASIMTUFII pre-filled syringe if the suspension is discolored, or particulate matter is present.
- One sterile 1 ½ inch 22 gauge needle (in black packaging)
- One sterile 2 inch 21 gauge needle (in green packaging)
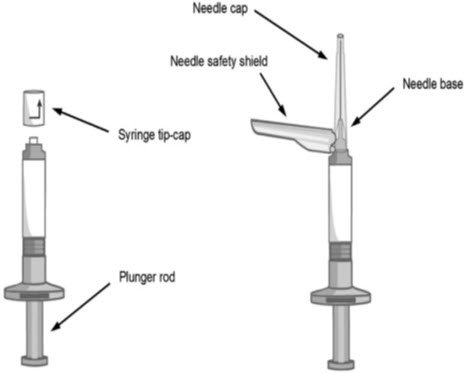
- Remove the ABILIFY ASIMTUFII pre-filled syringe from the package.
- Tap the syringe on your hand at least 10 (ten) times (Figure 1).
- After tapping, shake the syringe vigorously for at least 10 (ten) seconds until the medication is uniform (Figure 2).
- For non-obese patients - 22-gauge, 1.5-inch (38 mm) safety needle with needle protection device (needle in black packaging)
- For obese patients - 21-gauge, 2-inch (51 mm) safety needle with needle protection device (needle in green packaging)
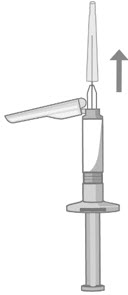
- Twist and pull off the pre-filled syringe tip-cap (Figure 3).
- While holding the base of the needle, ensure the needle is firmly seated on the safety device with a push. Gently twist clockwise until SECURELY fitted (Figure 3).

- When you are ready to administer the injection of ABILIFY ASIMTUFII, hold the pre-filled syringe upright and remove the needle-cap straight up (Figure 4). Do not twist the needle-cap, as this may loosen the needle from the syringe.
- Slowly advance the plunger rod upward to expel the air and until the suspension fills needle base (Figure 5).
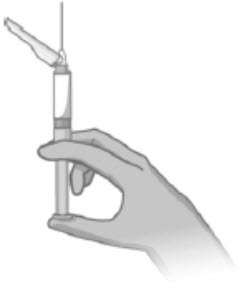
- Slowly inject the entire contents of the pre-filled syringe intramuscularly into the gluteal muscle of the patient (Figure 6).
- After the injection, press the safety shield on a hard surface to cover and lock shield over the needle (Figure 7 and 8)
- Immediately discard used syringe and the unused needle in an approved sharps container (Figure 9).
- The unused needle should not be saved for future use.



