Generic Name
Ultramicrosize
Brand Names
Grisofulvin, Microsize, Fulvicin
FDA approval date: September 09, 1971
Classification: Tubulin Inhibiting Agent
Form: Tablet, Suspension
What is Grisofulvin (Ultramicrosize)?
Ultramicrosize griseofulvin tablets are indicated for the treatment of the following ringworm infections; tinea corporis , tinea pedis , tinea cruris , tinea barbae , tinea capitis , and tinea unguium , when caused by one or more of the following genera of fungi: Trichophyton rubrum, Trichophyton tonsurans, Trichophyton mentagrophytes, Trichophyton interdigitalis,Trichophyton verrucosum, Trichophyton megnini, Trichophyton gallinae, Trichophyton crateriform, Trichophyton sulphureum, Trichophyton schoenleini, Microsporum audouini, Microsporum canis, Microsporum gypseum and Epidermophyton floccosum. NOTE: Prior to therapy, the type of fungi responsible for the infection should be identified. The use of the drug is not justified in minor or trivial infections which will respond to topical agents alone. Griseofulvin is not effective in the following: bacterial infections, candidiasis , histoplasmosis, actinomycosis, sporotrichosis, chromoblastomycosis, coccidioidomycosis, North American blastomycosis, cryptococcosis , tinea versicolor and nocardiosis.
Approved To Treat
Top Global Experts
There are no experts for this drug
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
There is no clinical trials being done for this treatment
Related Latest Advances
Brand Information
grisofulvin (grisofulvin)
1DESCRIPTION
Griseofulvin oral suspension USP (microsize) contains griseofulvin, USP (microsize) for oral administration. The active ingredient, griseofulvin, USP, is a fungistatic antibiotic, derived from a species of
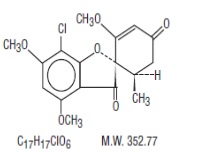
Griseofulvin, USP occurs as a white to creamy white, odorless powder which is very slightly soluble in water, soluble in acetone, dimethylformamide, and chloroform and sparingly soluble in alcohol.
Griseofulvin is a pink to orange colored, tutti-frutti flavored, uniform suspension. Each 5 mL of griseofulvin suspension contains 125 mg of griseofulvin, USP (microsize) and also contains the following inactive ingredients: artificial flavors, carboxymethyl cellulose sodium, dl-alpha-tocopherol, FD&C red #40, FD&C yellow #6, glycerin, hydrochloric acid, methylparaben, microcrystalline cellulose, propylene glycol, propylparaben, simethicone emulsion, sodium alginate, sodium hydroxide, sodium lauryl sulfate and sucrose.
2CLINICAL PHARMACOLOGY
Griseofulvin absorption from the gastrointestinal tract varies considerably among individuals mainly because of insolubility of the drug in aqueous media of the upper GI tract. Drug absorption has been estimated to range between 27 and 72%. After an oral dose, griseofulvin is primarily absorbed from the duodenum with some absorption occurring from the jejunum and ileum. The peak serum level in fasting adults given 0.5 g of griseofulvin microsize occurs at about four hours and ranges between 0.5 to 2 mcg/mL. The serum level may be increased by giving the drug with a meal with a high fat content. In one study in pediatric patients 19 months to 11 years of age, 10 mg/kg of griseofulvin microsize given with milk resulted in mean peak serum concentrations approximately four-fold greater than the same griseofulvin dose given alone (1.29 mcg/mL versus 0.34 mcg/mL, respectively). Also, the area under the curve value was ten-fold larger when 10 mg/kg griseofulvin and milk were administered simultaneously as compared to the same dosage given to fasting patients. In addition, griseofulvin administered with milk resulted in more consistently detected serum levels across subjects.
Following oral administration, griseofulvin is deposited in the keratin precursor cells and has a greater affinity for diseased tissue. The drug is tightly bound to the new keratin which becomes highly resistant to fungal invasions. When the drug is discontinued, griseofulvin concentrations in the skin decline less rapidly than those in plasma.
Griseofulvin is metabolized by the liver to 6-desmethylgriseofulvin and its glucuronide conjugate.
Griseofulvin has a variable elimination half-life in plasma (9 to 24 hours). Approximately 30% of a single oral dose of griseofulvin is excreted in the urine within 24 hours and about 50% of the dose is excreted in the urine within 5 days, mostly in the form of metabolites. Unchanged griseofulvin in the urine accounts for less than 1% of the administered dose. In addition, approximately one-third of a single dose of griseofulvin is excreted in feces within 5 days. Griseofulvin is also excreted in perspiration.
Microbiology
Mechanism of Action
The mechanism of griseofulvin consists of binding microtubular proteins, which are required for mitosis.
Activity
Griseofulvin may be active against most strains of the following dermatophytes as described in the
Epidermophyton floccosum, Microsporum audouinii, Microsporum canis, Microsporum gypseum, Trichophyton crateriformis, Trichophyton gallinae, Trichophyton interdigitalis, Trichophyton megnini, Trichophyton mentagrophytes, Trichophyton rubrum, Trichophyton sulphureum, Trichophyton schoenleini, Trichophyton tonsurans, and Trichophyton verrucosum.
It has no effect on bacteria or on other genera of fungi.
Activity
In vitro, griseofulvin has been shown to have activity against many dermatophytes, but the clinical significance is unknown.
Drug Resistance
Although there have been reports of dermatophyte resistance to griseofulvin, the clinical significance is unknown.
3INDICATIONS AND USAGE
Griseofulvin oral suspension is indicated for the treatment of dermatophyte infections of the skin not adequately treated by topical therapy, hair and nails, namely:
Tinea corporis
Tinea pedis
Tinea cruris
Tinea barbae
Tinea capitis
Tinea unguium when caused by one or more of the following species of fungi:
Epidermophyton floccosum
Microsporum audouinii
Microsporum canis
Microsporum gypseum
Trichophyton crateriformis
Trichophyton gallinae
Trichophyton interdigitalis
Trichophyton megnini
Trichophyton mentagrophytes
Trichophyton rubrum
Trichophyton schoenleini
Trichophyton sulphureum
Trichophyton tonsurans
Trichophyton verrucosum
Note: Prior to therapy, a dermatophyte should be identified as responsible for the infection.
Prior to initiating treatment, appropriate specimens for laboratory testing (KOH preparation, fungal culture, or nail biopsy) should be obtained to confirm the diagnosis.
Griseofulvin is
Bacterial infections Coccidioidomycosis
Candidiasis (Moniliasis) North American Blastomycosis
Histoplasmosis Cryptococcosis (Torulosis)
Actinomycosis Tinea versicolor
Sporotrichosis Nocardiosis
Chromoblastomycosis
The use of this drug is not justified in minor or trivial dermatophyte infections which will respond to topical agents alone.
4CONTRAINDICATIONS
Griseofulvin is contraindicated in patients with porphyria or hepatocellular failure, and in individuals with a history of hypersensitivity to griseofulvin.
Griseofulvin may cause fetal harm when administered to a pregnant woman. Two published cases of conjoined twins have been reported in patients taking griseofulvin during the first trimester of pregnancy, therefore, griseofulvin is contraindicated in women who are or may become pregnant during treatment. Women taking estrogen-containing oral contraceptives may be at increased risk of becoming pregnant while on griseofulvin (see also
Griseofulvin has been shown to be embryotoxic and teratogenic in pregnant rats when given at a daily oral dose of 250 mg/kg/day [4X the Maximum Recommended Human Dose (MRHD) based on Body Surface Area (BSA)]. Griseofulvin also has been shown to be embryotoxic and teratogenic in pregnant cats treated weekly with griseofulvin at doses of 500 to 1000 mg/week. There are reports of teratogenicity in a Golden Retriever when doses of 750 mg/day [1.2X the MRHD based on BSA] were administered for four weeks prior to and throughout the pregnancy, and in a study in which beagles were administered 35 mg/kg/day [1.9X the MRHD based on BSA] for intervals from one week up to the entire gestation period. Teratogenicity was also seen in mice when griseofulvin was administered in doses equivalent to 5 g/kg/day [40X the MRHD based on BSA] for 2 consecutive
days at various stages of the pregnancy.
5ADVERSE REACTIONS
There have been postmarketing reports of severe skin and hepatic adverse events associated with griseofulvin use (see
When adverse reactions occur, they are most commonly of the hypersensitivity type, such as skin rashes, urticaria, and rarely, angioneurotic edema, and erythema multiforme. These may necessitate withdrawal of therapy and appropriate countermeasures. Peripheral neuropathy and paresthesias of the hands and feet have been reported and may be related to treatment duration. Most patients treated with griseofulvin for less than six months experienced improvement or resolution of their neuropathy upon withdrawal of the griseofulvin. Other side effects reported occasionally are oral thrush, nausea, vomiting, epigastric distress, diarrhea, headache, fatigue, dizziness, insomnia, mental confusion, and impairment of performance of routine activities.
Proteinuria, nephrosis (sometimes associated with existing systemic lupus erythematosus), leukopenia, coagulopathy, hepatitis, elevated liver enzymes, hyperbilirubinemia, and GI bleeding have been reported rarely. Administration of the drug should be discontinued if granulocytopenia occurs.
6OVERDOSAGE
There is limited experience on overdose with griseofulvin. In case of overdosage, discontinue medication, treat symptomatically and institute supportive measures as required.
7DOSAGE AND ADMINISTRATION
Accurate diagnosis of the infecting organism is essential. Identification should be made either by direct microscopic examination of a mounting of infected tissue in a solution of potassium hydroxide or by culture on an appropriate medium.
Medication must be continued until the infecting organism is completely eradicated as indicated by appropriate clinical or laboratory examination. Representative treatment periods are tinea capitis, 4 to 6 weeks; tinea corporis, 2 to 4 weeks; tinea pedis, 4 to 8 weeks; tinea unguium – depending on rate of growth – fingernails, at least 4 months; toenails, at least 6 months.
General measures in regard to hygiene should be observed to control sources of infection or reinfection. Concomitant use of appropriate topical agents is usually required, particularly in treatment of tinea pedis. In some forms of tinea pedis, yeasts and bacteria may be involved as well as dermatophytes. Griseofulvin will not eradicate these associated bacterial or yeast infections.
ADULTS: 0.5 g daily (125 mg q.i.d., 250 mg b.i.d., or 500 mg/day). Patients with less severe or 300 extensive infections may require less, whereas those with widespread lesions may require a starting dose of 0.75 g to 1 g/day. This may be reduced gradually to 0.5 g or less after a response has been noted. In all cases, the dosage should be individualized.
PEDIATRIC PATIENTS (older than 2 years): A dosage of 10 mg/kg daily is usually adequate (pediatric patients from 30 to 50 lb, 125 mg to 250 mg daily; pediatric patients over 50 lb, 250 mg to 500 mg daily, in divided doses). Dosage should be individualized, as with adults. Clinical relapse will occur if the medication is not continued until the infecting organism is eradicated.
Safety is not established at higher doses than recommended.
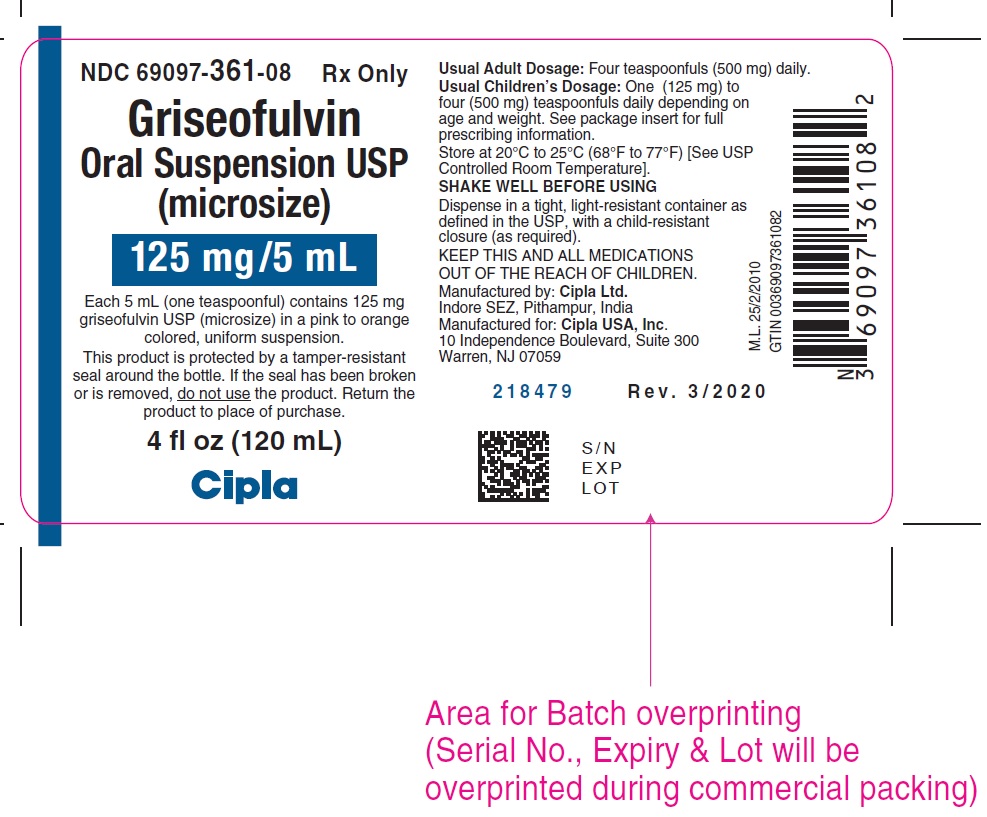
8HOW SUPPLIED
Griseofulvin Oral Suspension USP is available as a pink to orange colored, tutti-frutti flavored, uniform suspension containing 125 mg/5 mL griseofulvin, USP (microsize) in a 4 ounce (120 mL) bottle.
NDC- 69097-361-08
PHARMACIST: Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure as required.
This product is protected by a tamper-resistant seal around the bottle. If the seal has been broken or is removed,
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
SHAKE WELL BEFORE USING
Manufactured by:
Cipla Ltd., Indore SEZ, Pithampur, India
Manufactured for:
Cipla USA, Inc.
10 Independence Boulevard, Suite 300
Warren, NJ 07059
Revised: 3/2020
9PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 69097-361-08 RX only
Griseofulvin
Oral Suspension USP
(microsize)
125mg/5mL
Each 5 mL (one teaspoonful) contains 125 mg
griseofulvin USP (microsize) in a pink to orange
colored, uniform suspension.
4 fl oz (120 mL)
Cipla