Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of KENGREAL has been evaluated in 13,301 subjects in controlled trials, in whom, 5,529 were in the CHAMPION PHOENIX trial.
Bleeding
There was a greater incidence of bleeding with KENGREAL than with clopidogrel. No baseline demographic factor altered the relative risk of bleeding with KENGREAL (see Table 1 and Figure 1).
Abbreviations: GUSTO: Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Arteries; TIMI: Thrombolysis in Myocardial Infarction
a Safety population is all randomized subjects who received at least one dose of study drug
b intracranial hemorrhage or bleeding resulting in substantial hemodynamic compromise requiring treatment
c requiring blood transfusion but not resulting in hemodynamic compromise
d all other bleeding not included in severe or moderate
e any intracranial hemorrhage, or any overt bleeding associated with a reduction in hemoglobin of ≥5 g/dL (or, when hemoglobin is not available, an absolute reduction in hematocrit ≥15%)
f any overt sign of bleeding (including observation by imaging techniques) that is associated with a reduction in hemoglobin of ≥3 g/dL and <5 g/dL (or, when hemoglobin is not available, an absolute reduction in hematocrit of ≥9% and <15%)
Figure 1:Bleeding Results in the CHAMPION PHOENIX Studya (All Non-CABG related)
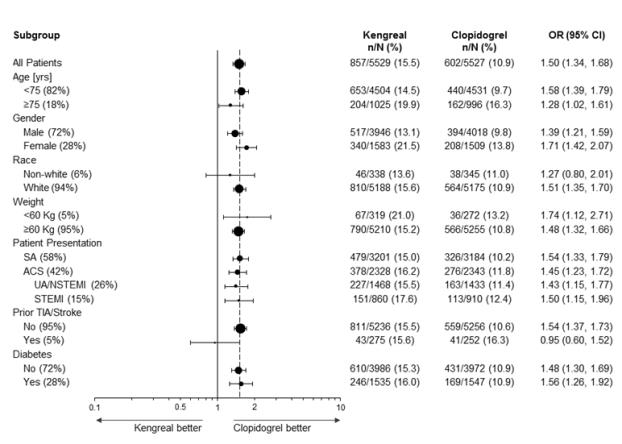
a Safety population is all randomized subjects who received at least one dose of study drug
Note: The figure above presents effects in various subgroups most of which are baseline characteristics and most of which were pre-specified (patient presentation and weight were not pre-specified subgroups). The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
Drug Discontinuation
In CHAMPION PHOENIX the rate of discontinuation for bleeding events was 0.3% for KENGREAL and 0.1% for clopidogrel. Discontinuation for non-bleeding adverse events was low and similar for KENGREAL (0.6%) and for clopidogrel (0.4%). Coronary artery dissection, coronary artery perforation, and dyspnea were the most frequent events leading to discontinuation in patients treated with KENGREAL.
Non-Bleeding Adverse Reactions
Hypersensitivity
Serious cases of hypersensitivity were more frequent with KENGREAL (7/13301) than with control (2/12861). These included anaphylactic reactions, anaphylactic shock, bronchospasm, angioedema, and stridor.
Decreased renal function
Worsening renal function was reported in 3.2% of KENGREAL patients with severe renal impairment (creatinine clearance <30 mL/min) compared to 1.4% of clopidogrel patients with severe renal impairment.
Dyspnea
Dyspnea was reported more frequently in patients treated with KENGREAL (1.3%) than with control (0.4%).