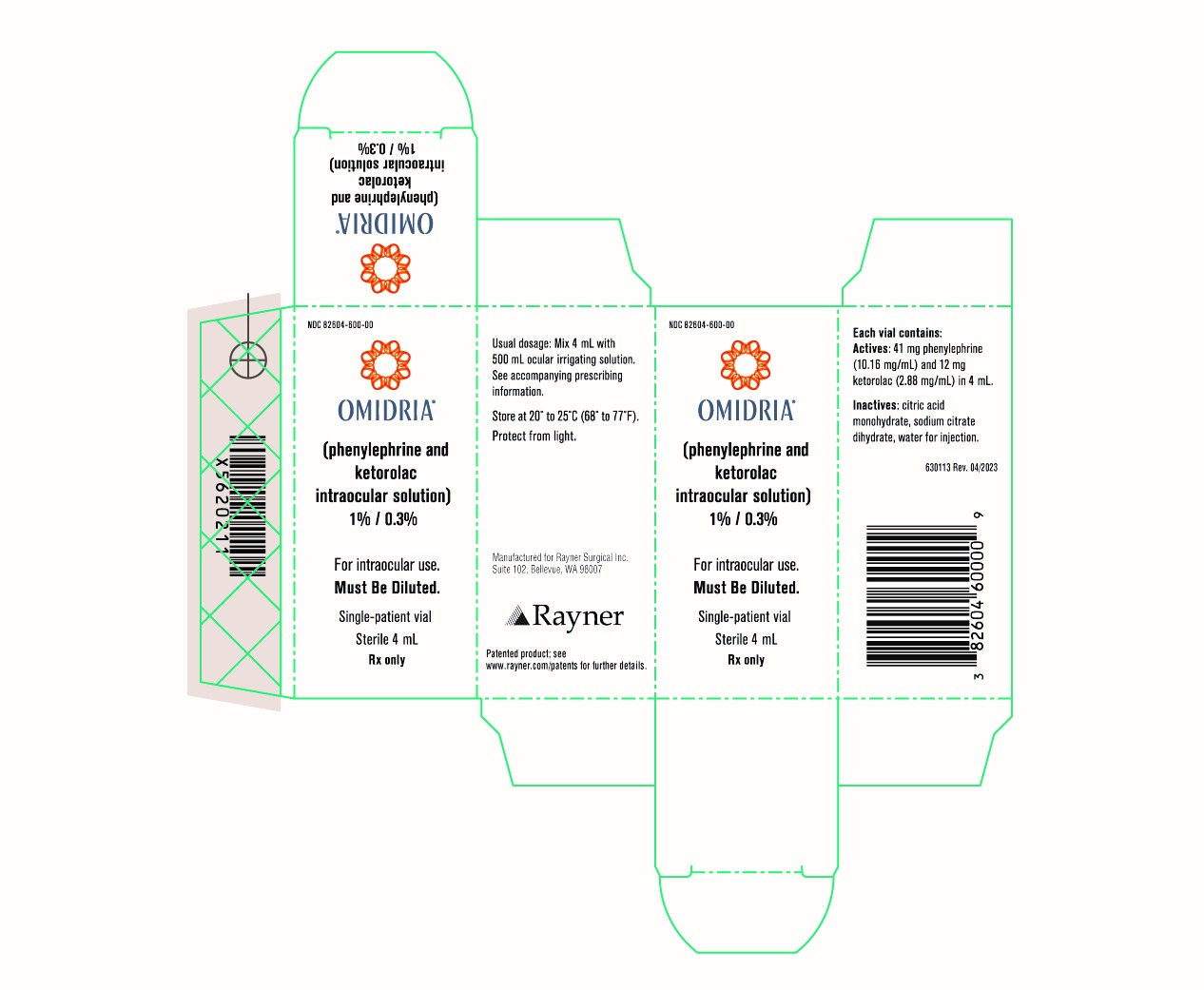
Omidria
What is Omidria (Ketorolac)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: This single-centre, randomized, double-blind, controlled clinical trial aims to evaluate the effectiveness of preoperative photobiomodulation (PBM) with red and infrared wavelengths in reducing periorbital edema in patients undergoing rhinoplasty. Sixty participants will be randomized to receive either active PBM or sham PBM one hour before surgery. The primary outcome is periorbital edema on post...
Summary: This is a prospective interventional study examining the effect of ketorolac at doses of 15mg versus 30 mg for duration of analgesia in emergency department patients with suspected renal colic.
Summary: The purpose of this study is to characterize the pharmacokinetic (PK) and safety profile of anesthetics and analgesics in children and adolescents.
Related Latest Advances
Brand Information
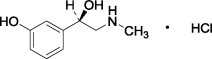
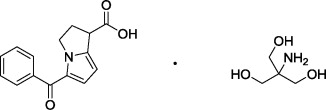
Patented product; see www.rayner.com/patents for further details.
640069