Tazobactam
What is Piperacillin (Tazobactam)?
Approved To Treat
Related Clinical Trials
Objectives: 1. To determine the safety and efficacy of a local Chlorin E6-mediated Photodynamic Therapy in patients with diabetic foot ulcers. 2. To determine the effect of Chlorin E6 on the healing of diabetic foot ulcers defined by a reduction in size and depth of the ulcer. 3. To compare the efficacy and healing rates of Diabetic foot ulcers with and without local Chlorin E6-mediated Photodynamic Therapy a...
Summary: The goal of this clinical trial is to learn whether subcutaneous piperacillin/tazobactam can provide drug exposure similar to standard intravenous piperacillin/tazobactam in adults who need treatment for an infection. It will also assess safety and clinical outcomes. The main questions it aims to answer are: * Does subcutaneous piperacillin/tazobactam achieve pharmacokinetic exposure comparable to...
Summary: Necrotizing soft tissue infection (NSTI) is a devastating disease that results in a high rate of in-hospital complications and despite advances in critical care, wound care, and early intervention, NSTI continues to be associated with a mortality rate of nearly 30%. The antibiotics used in this treatment are Clindamycin, Vancomycin, Piperacillin Tazobactam; these antibiotics may be administered co...
Related Latest Advances
Brand Information
- Hypersensitivity Adverse Reactions
- Severe Cutaneous Adverse Reactions
- Hemophagocytic Lymphohistiocytosis
- Rhabdomyolysis [
- Hematologic Adverse Reactions
- Central Nervous System Adverse Reactions
- Nephrotoxicity in Critically Ill Patients
- Clostridioides difficile-Associated Diarrhea [see Warnings and Precautions (5.9)]
During the initial clinical investigations, 2621 patients worldwide were treated with piperacillin and tazobactam in phase 3 trials. In the key North American monotherapy clinical trials (n=830 patients), 90% of the adverse events reported were mild to moderate in severity and transient in nature. However, in 3.2% of the patients treated worldwide, piperacillin and tazobactam was discontinued because of adverse events primarily involving the skin (1.3%), including rash and pruritus; the gastrointestinal system (0.9%), including diarrhea, nausea, and vomiting; and allergic reactions (0.5%).
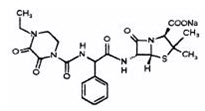
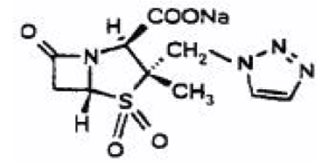
- Pharmacy Bulk Bottle
- Each pharmacy bulk package bottle provides piperacillin sodium equivalent to 36 grams of piperacillin and tazobactam sodium equivalent to 4.5 grams of tazobactam. Each pharmacy bulk package bottle contains 84.6 mEq (1,945 mg) of sodium.

PIPERACILLIN AND TAZOBACTAM
Pharmacy Bulk Package-
Rx only




