Brand Name
Eptifibatide
View Brand InformationFDA approval date: December 08, 2015
Classification: Platelet Aggregation Inhibitor
Form: Injection
What is Eptifibatide?
Eptifibatide Injection is a platelet aggregation inhibitor indicated for: Treatment of acute coronary syndrome managed medically or with percutaneous coronary intervention .
Approved To Treat
Top Global Experts
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
There is no clinical trials being done for this treatment
Related Latest Advances
Brand Information
Eptifibatide (eptifibatide)
1DOSAGE AND ADMINISTRATION
Before infusion of eptifibatide injection, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured.
The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
1.1Dosage in Acute Coronary Syndrome (ACS)
Eptifibatide injection should be given concomitantly with heparin dosed to achieve the following parameters:
During Medical Management: Target aPTT 50 to 70 seconds
- If weight greater than or equal to 70 kg, 5000-unit bolus followed by infusion of 1000 units/h.
- If weight less than 70 kg, 60-units/kg bolus followed by infusion of 12 units/kg/h.
During PCI: Target ACT 200 to 300 seconds
- If heparin is initiated prior to PCI, additional boluses during PCI to maintain an ACT target of 200 to 300 seconds.
- Heparin infusion after the PCI is discouraged.
1.2Dosage in Percutaneous Coronary Intervention (PCI)
- Eptifibatide injection should be given concomitantly with heparin to achieve a target ACT of 200 to 300 seconds. Administer 60-units/kg bolus initially in patients not treated with heparin within 6 hours prior to PCI.
- Additional boluses during PCI to maintain ACT within target.
- Heparin infusion after the PCI is strongly discouraged.
Patients requiring thrombolytic therapy should discontinue eptifibatide injection.
1.3Important Administration Instructions
- Inspect eptifibatide injection for particulate matter and discoloration prior to administration, whenever solution and container permit.
- May administer eptifibatide injection in the same intravenous line as alteplase, atropine, dobutamine, heparin, lidocaine, meperidine, metoprolol, midazolam, morphine, nitroglycerin, or verapamil. Do not administer eptifibatide injection through the same intravenous line as furosemide.
- May administer eptifibatide injection in the same IV line with 0.9% NaCl or 0.9% NaCl/5% dextrose. With either vehicle, the infusion may also contain up to 60 mEq/L of potassium chloride.
- Withdraw the bolus dose(s) of eptifibatide injection from the 10 mL vial into a syringe. Administer the bolus dose(s) by IV push.
- Immediately following the bolus dose administration, initiate a continuous infusion of eptifibatide injection. When using an intravenous infusion pump, administer eptifibatide injection undiluted directly from the 100-mL vial. Spike the 100 mL vial with a vented infusion set. Center the spike within the circle on the stopper top.
- Discard any unused portion left in the vial.
Administer eptifibatide injection by volume according to patient weight (see
2DOSAGE FORMS AND STRENGTHS
- Injection: 20 mg of eptifibatide injection in 10 mL (2 mg/mL), for intravenous bolus.
- Injection: 75 mg of eptifibatide injection in 100 mL (0.75 mg/mL), for intravenous infusion.
3CONTRAINDICATIONS
Treatment with eptifibatide injection is contraindicated in patients with:
- A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days
- Severe hypertension (systolic blood pressure >200 mm Hg or diastolic blood pressure >110 mm Hg) not adequately controlled on antihypertensive therapy
- Major surgery within the preceding 6 weeks
- History of stroke within 30 days or any history of hemorrhagic stroke
- Current or planned administration of another parenteral GP IIb/IIIa inhibitor
- Dependency on renal dialysis
- Hypersensitivity to eptifibatide injection or any component of the product (hypersensitivity reactions that occurred included anaphylaxis and urticaria).
4ADVERSE REACTIONS
The following serious adverse reaction is also discussed elsewhere in the labeling:
- Bleeding
4.1Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
A total of 16,782 patients were treated in the Phase III clinical trials (PURSUIT, ESPRIT, and IMPACT II)
Bleeding and hypotension were the most commonly reported adverse reactions (incidence ≥5% and greater than placebo) in the eptifibatide injection controlled clinical trial database.
Bleeding
The incidence of bleeding and transfusions in the PURSUIT and ESPRIT studies are shown in
The majority of major bleeding reactions in the ESPRIT study occurred at the vascular access site (1 and 8 patients, or 0.1% and 0.8% in the placebo and eptifibatide injection groups, respectively). Bleeding at “other” locations occurred in 0.2% and 0.4% of patients, respectively.
In the PURSUIT study, the greatest increase in major bleeding in eptifibatide-treated patients compared to placebo-treated patients was also associated with bleeding at the femoral artery access site (2.8% versus 1.3%). Oropharyngeal (primarily gingival), genitourinary, gastrointestinal, and retroperitoneal bleeding were also seen more commonly in eptifibatide-treated patients compared to placebo-treated patients.
Among patients experiencing a major bleed in the IMPACT II study, an increase in bleeding on eptifibatide injection versus placebo was observed only for the femoral artery access site (3.2% versus 2.8%).
Table 3 displays the incidence of TIMI major bleeding according to the cardiac procedures carried out in the PURSUIT study. The most common bleeding complications were related to cardiac revascularization (CABG-related or femoral artery access site bleeding). A corresponding table for ESPRIT is not presented, as every patient underwent PCI in the ESPRIT study and only 11 patients underwent CABG.
In the PURSUIT and ESPRIT studies, the risk of major bleeding with eptifibatide injection increased as patient weight decreased. This relationship was most apparent for patients weighing less than 70 kg.
Bleeding resulting in discontinuation of the study drug was more frequent among patients receiving eptifibatide injection than placebo (4.6% versus 0.9% in ESPRIT, 8% versus 1% in PURSUIT, 3.5% versus 1.9% in IMPACT II).
Intracranial Hemorrhage and Stroke
Intracranial hemorrhage was rare in the PURSUIT, IMPACT II, and ESPRIT clinical studies. In the PURSUIT study, 3 patients in the placebo group, 1 patient in the group treated with eptifibatide injection 180/1.3, and 5 patients in the group treated with eptifibatide injection 180/2 experienced a hemorrhagic stroke. The overall incidence of stroke was 0.5% in patients receiving eptifibatide injection 180/1.3, 0.7% in patients receiving eptifibatide injection 180/2, and 0.8% in placebo patients.
In the IMPACT II study, intracranial hemorrhage was experienced by 1 patient treated with eptifibatide injection 135/0.5, 2 patients treated with eptifibatide injection 135/0.75, and 2 patients in the placebo group. The overall incidence of stroke was 0.5% in patients receiving 135/0.5 eptifibatide injection, 0.7% in patients receiving eptifibatide injection 135/0.75, and 0.7% in the placebo group.
In the ESPRIT study, there were 3 hemorrhagic strokes, 1 in the placebo group and 2 in the eptifibatide injection group. In addition there was 1 case of cerebral infarction in the eptifibatide injection group.
Immunogenicity/Thrombocytopenia
The potential for development of antibodies to eptifibatide has been studied in 433 subjects. Eptifibatide injection was nonantigenic in 412 patients receiving a single administration of eptifibatide injection (135 mcg/kg bolus followed by a continuous infusion of either 0.5 mcg/kg/min or 0.75 mcg/kg/min), and in 21 subjects to whom eptifibatide injection (135 mcg/kg bolus followed by a continuous infusion of 0.75 mcg/kg/min) was administered twice, 28 days apart. In both cases, plasma for antibody detection was collected approximately 30 days after each dose. The development of antibodies to eptifibatide at higher doses has not been evaluated.
In patients with suspected eptifibatide-related immune-mediated thrombocytopenia, IgG antibodies that react with the GP IIb/IIIa complex were identified in the presence of eptifibatide and in eptifibatide-naïve patients. These findings suggest acute thrombocytopenia after the administration of eptifibatide injection can develop as a result of naturally occurring drug-dependent antibodies or those induced by prior exposure to eptifibatide injection. Similar antibodies were identified with other GP IIb/IIIa ligand-mimetic agents. Immune-mediated thrombocytopenia with eptifibatide injection may be associated with hypotension and/or other signs of hypersensitivity.
In the PURSUIT and IMPACT II studies, the incidence of thrombocytopenia (<100,000/mm
Other Adverse Reactions
In the PURSUIT and ESPRIT studies, the incidence of serious nonbleeding adverse reactions was similar in patients receiving placebo or eptifibatide injection (19% and 19%, respectively, in PURSUIT; 6% and 7%, respectively, in ESPRIT). In PURSUIT, the only serious nonbleeding adverse reaction that occurred at a rate of at least 1% and was more common with eptifibatide injection than placebo (7% versus 6%) was hypotension. Most of the serious nonbleeding adverse reactions consisted of cardiovascular reactions typical of a UA population. In the IMPACT II study, serious nonbleeding adverse reactions that occurred in greater than 1% of patients were uncommon and similar in incidence between placebo- and eptifibatide-treated patients.
Discontinuation of study drug due to adverse reactions other than bleeding was uncommon in the PURSUIT, IMPACT II, and ESPRIT studies, with no single reaction occurring in >0.5% of the study population (except for “other” in the ESPRIT study).
4.2Postmarketing Experience
The following adverse reactions have been reported in post-approval use of eptifibatide injection in combination with heparin and aspirin. Because the reactions below are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure: cerebral, GI, and pulmonary hemorrhage. Fatal bleeding reactions have been reported. Acute profound thrombocytopenia, as well as immune-mediated thrombocytopenia, has been reported
5OVERDOSAGE
There has been only limited experience with overdosage of eptifibatide injection. There were 8 patients in the IMPACT II study, 9 patients in the PURSUIT study, and no patients in the ESPRIT study who received bolus doses and/or infusion doses more than double those called for in the protocols. None of these patients experienced an intracranial bleed or other major bleeding.
Eptifibatide was not lethal to rats, rabbits, or monkeys when administered by continuous intravenous infusion for 90 minutes at a total dose of 45 mg/kg (about 2 to 5 times the recommended maximum daily human dose on a body surface area basis). Symptoms of acute toxicity were loss of righting reflex, dyspnea, ptosis, and decreased muscle tone in rabbits and petechial hemorrhages in the femoral and abdominal areas of monkeys.
From
6DESCRIPTION
Eptifibatide is a cyclic heptapeptide containing 6 amino acids and 1 mercaptopropionyl (des-amino cysteinyl) residue. An interchain disulfide bridge is formed between the cysteine amide and the mercaptopropionyl moieties. Chemically it is N
The eptifibatide peptide is produced by solution-phase peptide synthesis, and is purified by preparative reverse-phase liquid chromatography and lyophilized. The structural formula is:
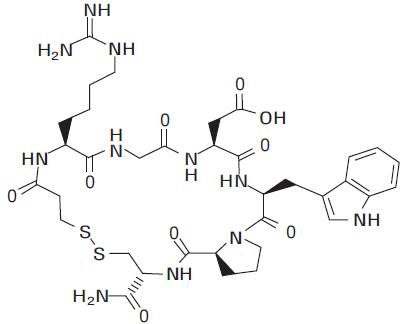
Eptifibatide Injection is a clear, colorless, sterile, non-pyrogenic solution for intravenous (IV) use with an empirical formula of C
7CLINICAL STUDIES
Eptifibatide injection was studied in 3 placebo-controlled, randomized studies. PURSUIT evaluated patients with acute coronary syndromes: UA or NSTEMI. Two other studies, ESPRIT and IMPACT II, evaluated patients about to undergo a PCI. Patients underwent primarily balloon angioplasty in IMPACT II and intracoronary stent placement, with or without angioplasty, in ESPRIT.
7.1Non-ST-Segment Elevation Acute Coronary Syndrome
Non-ST-segment elevation acute coronary syndrome is defined as prolonged (≥10 minutes) symptoms of cardiac ischemia within the previous 24 hours associated with either ST-segment changes (elevations between 0.6 mm and 1 mm or depression >0.5 mm), T-wave inversion (>1 mm), or positive CK-MB. This definition includes “unstable angina” and “NSTEMI” but excludes MI that is associated with Q waves or greater degrees of ST-segment elevation.
PURSUIT (Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Eptifibatide Injection Therapy)
PURSUIT was a 726-center, 27-country, double-blind, randomized, placebo-controlled study in 10,948 patients presenting with UA or NSTEMI. Patients could be enrolled only if they had experienced cardiac ischemia at rest (≥10 minutes) within the previous 24 hours and had either ST-segment changes (elevations between 0.6 mm and 1 mm or depression >0.5 mm), T-wave inversion (>1 mm), or increased CK-MB. Important exclusion criteria included a history of bleeding diathesis, evidence of abnormal bleeding within the previous 30 days, uncontrolled hypertension, major surgery within the previous 6 weeks, stroke within the previous 30 days, any history of hemorrhagic stroke, serum creatinine >2 mg/dL, dependency on renal dialysis, or platelet count <100,000/mm
Patients were randomized to placebo, to eptifibatide injection 180 mcg/kg bolus followed by a 2 mcg/kg/min infusion (180/2), or to eptifibatide injection 180 mcg/kg bolus followed by a 1.3 mcg/kg/min infusion (180/1.3). The infusion was continued for 72 hours, until hospital discharge, or until the time of CABG, whichever occurred first, except that if PCI was performed, the eptifibatide infusion was continued for 24 hours after the procedure, allowing for a duration of infusion up to 96 hours.
The lower-infusion-rate arm was stopped after the first interim analysis when the 2 active-treatment arms appeared to have the same incidence of bleeding.
Patient age ranged from 20 to 94 (mean 63) years, and 65% were male. The patients were 89% Caucasian, 6% Hispanic, and 5% Black, recruited in the United States and Canada (40%), Western Europe (39%), Eastern Europe (16%), and Latin America (5%).
This was a “real world” study; each patient was managed according to the usual standards of the investigational site; frequencies of angiography, PCI, and CABG therefore differed widely from site to site and from country to country. Of the patients in PURSUIT, 13% were managed with PCI during drug infusion, of whom 50% received intracoronary stents; 87% were managed medically (without PCI during drug infusion).
The majority of patients received aspirin (75-325 mg once daily). Heparin was administered intravenously or subcutaneously, at the physician's discretion, most commonly as an intravenous bolus of 5000 units followed by a continuous infusion of 1000 units/h. For patients weighing less than 70 kg, the recommended heparin bolus dose was 60 units/kg followed by a continuous infusion of 12 units/kg/h. A target aPTT of 50 to 70 seconds was recommended. A total of 1250 patients underwent PCI within 72 hours after randomization, in which case they received intravenous heparin to maintain an ACT of 300 to 350 seconds.
The primary endpoint of the study was the occurrence of death from any cause or new MI (evaluated by a blinded Clinical Endpoints Committee) within 30 days of randomization.
Compared to placebo, eptifibatide injection administered as a 180 mcg/kg bolus followed by a 2 mcg/kg/min infusion significantly (p=0.042) reduced the incidence of endpoint events (see
Figure 1: Kaplan-Meier Plot of Time to Death or Myocardial Infarction Within 30 Days of Randomization in the PURSUIT Study
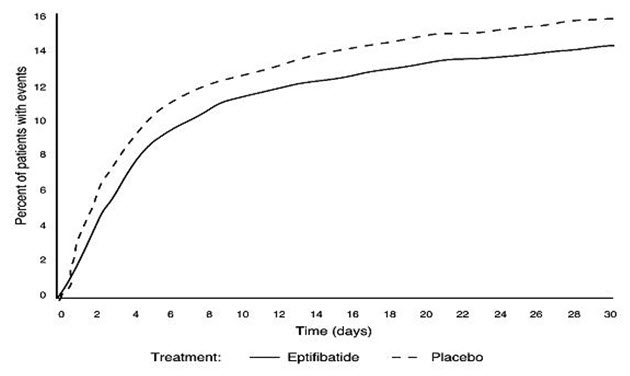
Treatment with eptifibatide injection prior to determination of patient management strategy reduced clinical events regardless of whether patients ultimately underwent diagnostic catheterization, revascularization (i.e., PCI or CABG surgery) or continued to receive medical management alone.
All of the effect of eptifibatide injection was established within 72 hours (during the period of drug infusion), regardless of management strategy. Moreover, for patients undergoing early PCI, a reduction in events was evident prior to the procedure.
An analysis of the results by sex suggests that women who would not routinely be expected to undergo PCI receive less benefit from eptifibatide injection (95% confidence limits for relative risk of 0.94 - 1.28) than do men (0.72 - 0.9). This difference may be a true treatment difference, the effect of other differences in these subgroups, or a statistical anomaly. No differential outcomes were seen between male and female patients undergoing PCI
Follow-up data were available through 165 days for 10,611 patients enrolled in the PURSUIT trial (96.9% of the initial enrollment). This follow-up included 4566 patients who received eptifibatide injection at the 180/2 dose. As reported by the investigators, the occurrence of death from any cause or new MI for patients followed for at least 165 days was reduced from 13.6% with placebo to 12.1% with eptifibatide injection 180/2.
7.2Percutaneous Coronary Intervention (PCI)
IMPACT II (Eptifibatide Injection to Minimize Platelet Aggregation and Prevent Coronary Thrombosis II)
IMPACT II was a multicenter, double-blind, randomized, placebo-controlled study conducted in the United States in 4010 patients undergoing PCI. Major exclusion criteria included a history of bleeding diathesis, major surgery within 6 weeks of treatment, gastrointestinal bleeding within 30 days, any stroke or structural CNS abnormality, uncontrolled hypertension, PT >1.2 times control, hematocrit <30%, platelet count <100,000/mm
Patient age ranged from 24 to 89 (mean 60) years, and 75% were male. The patients were 92% Caucasian, 5% Black, and 3% Hispanic. Forty-one percent of the patients underwent PCI for ongoing ACS. Patients were randomly assigned to 1 of 3 treatment regimens, each incorporating a bolus dose initiated immediately prior to PCI followed by a continuous infusion lasting 20 to 24 hours:
1) 135 mcg/kg bolus followed by a continuous infusion of 0.5 mcg/kg/min of eptifibatide injection (135/0.5);
2) 135 mcg/kg bolus followed by a continuous infusion of 0.75 mcg/kg/min of eptifibatide injection (135/0.75); or
3) a matching placebo bolus followed by a matching placebo continuous infusion.
Each patient received aspirin and an intravenous heparin bolus of 100 units/kg, with additional bolus infusions of up to 2000 additional units of heparin every 15 minutes to maintain an ACT of 300 to 350 seconds.
The primary endpoint was the composite of death, MI, or urgent revascularization, analyzed at 30 days after randomization in all patients who received at least 1 dose of study drug.
As shown in
ESPRIT (Enhanced Suppression of the Platelet IIb/IIIa Receptor with Eptifibatide Injection Therapy)
The ESPRIT study was a multicenter, double-blind, randomized, placebo-controlled study conducted in the United States and Canada that enrolled 2064 patients undergoing elective or urgent PCI with intended intracoronary stent placement. Exclusion criteria included MI within the previous 24 hours, ongoing chest pain, administration of any oral antiplatelet or oral anticoagulant other than aspirin within 30 days of PCI (although loading doses of thienopyridine on the day of PCI were encouraged), planned PCI of a saphenous vein graft or subsequent “staged” PCI, prior stent placement in the target lesion, PCI within the previous 90 days, a history of bleeding diathesis, major surgery within 6 weeks of treatment, gastrointestinal bleeding within 30 days, any stroke or structural CNS abnormality, uncontrolled hypertension, PT >1.2 times control, hematocrit <30%, platelet count <100,000/mm
Patient age ranged from 24 to 93 (mean 62) years, and 73% of patients were male. The study enrolled 90% Caucasian, 5% African American, 2% Hispanic, and 1% Asian patients. Patients received a wide variety of stents. Patients were randomized either to placebo or eptifibatide injection administered as an intravenous bolus of 180 mcg/kg followed immediately by a continuous infusion of 2 mcg/kg/min, and a second bolus of 180 mcg/kg administered 10 minutes later (180/2/180). Eptifibatide infusion was continued for 18 to 24 hours after PCI or until hospital discharge, whichever came first. Each patient received at least 1 dose of aspirin (162-325 mg) and 60 units/kg of heparin as a bolus (not to exceed 6000 units) if not already receiving a heparin infusion. Additional boluses of heparin (10-40 units/kg) could be administered in order to reach a target ACT between 200 and 300 seconds.
The primary endpoint of the ESPRIT study was the composite of death, MI, urgent target vessel revascularization (UTVR), and “bailout” to open-label eptifibatide injection due to a thrombotic complication of PCI (TBO) (e.g., visible thrombus, “no reflow,” or abrupt closure) at 48 hours. MI, UTVR, and TBO were evaluated by a blinded Clinical Events Committee.
As shown in
The need for thrombotic “bailout” was significantly reduced with eptifibatide injection at 48 hours (2.1% for placebo, 1% for eptifibatide injection; p=0.029). Consistent with previous studies of GP IIb/IIIa inhibitors, most of the benefit achieved acutely with eptifibatide injection was in the reduction of MI. Eptifibatide injection reduced the occurrence of MI at 48 hours from 9% for placebo to 5.4% (p=0.0015) and maintained that effect with significance at 30 days.
There was no treatment difference with respect to sex in ESPRIT. Eptifibatide injection reduced the incidence of the primary endpoint in both men (95% confidence limits for relative risk: 0.54, 1.07) and women (0.24, 0.72) at 48 hours.
Follow-up (12-month) mortality data were available for 2024 patients (1017 on eptifibatide injection) enrolled in the ESPRIT trial (98.1% of the initial enrollment). Twelve-month clinical event data were available for 1964 patients (988 on eptifibatide injection), representing 95.2% of the initial enrollment. As shown in
8PATIENT COUNSELING INFORMATION
Advise the patient to inform the doctor or healthcare provider about any medical conditions, medications, and allergies.
Rx only
Manufactured by:
Hainan Poly Pharm. Co., Ltd.
Distributed by:
Slate Run Pharmaceuticals, LLC
Columbus, Ohio 43215
10000060/04
Revised: 04/2021
9PRINCIPAL DISPLAY PANEL - 20 mg CARTON LABEL
NDC 70436-026-80
Eptifibatide Injection, 20 mg/10 mL (2 mg/mL)
10PRINCIPAL DISPLAY PANEL - 75 mg CARTON LABEL
NDC 70436-027-80
Eptifibatide Injection, 75 mg/100 mL (0.75 mg/mL)

