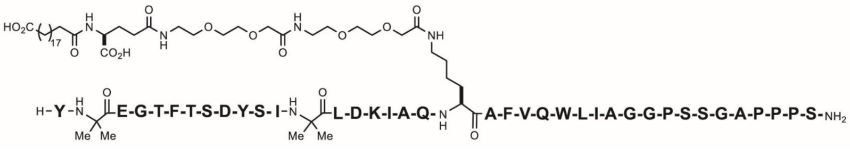
Tirzepatide
What is Zepbound (Tirzepatide)?
Approved To Treat
Related Clinical Trials
Summary: This trial aims to asses if tirzepatide-induced weight loss will lead to metabolic and hormonal changes in hormone receptor-positive (HR+), human epidermal growth factor receptor-negative (HER2-), node-positive (N+) high risk early breast cancer patients with obesity or overweight, inhibiting the growth and survival of micrometastatic disease and leading to clearance of tumor-informed circulating ...
Summary: The main purpose of this study is to find out how well and how safely tirzepatide works in adults who have type 1 diabetes and obesity or overweight. Participation in the study will last about 49 weeks.
Summary: The main purpose of the SYNERGY-OUTCOMES study is to find out whether retatrutide and tirzepatide can prevent major adverse liver outcomes (MALO) in people with high-risk metabolic dysfunction-associated steatotic liver disease (MASLD). The study will enroll adults who have MASLD based on non-invasive tests (NITs), which indicate they are more likely to develop MALO. Participants will be randomly ...
Related Latest Advances
Brand Information
- In rats, tirzepatide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether ZEPBOUND causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of tirzepatide-induced rodent thyroid C-cell tumors has not been determined[see Warnings and Precautions (
- ZEPBOUND is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)[see Contraindications (. Counsel patients regarding the potential risk for MTC with the use of ZEPBOUND and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Routine monitoring of serum calcitonin or using thyroid ultrasound is of uncertain value for early detection of MTC in patients treated with ZEPBOUND[see Contraindications (
- to reduce excess body weight and maintain weight reduction long term in adults with obesity or adults with overweight in the presence of at least one weight-related comorbid condition.
- to treat moderate to severe obstructive sleep apnea (OSA) in adults with obesity.
- A personal or family history of MTC or in patients with MEN 2
- Known serious hypersensitivity to tirzepatide or any of the excipients in ZEPBOUND. Serious hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with tirzepatide
- Risk of Thyroid C-cell Tumors
- Severe Gastrointestinal Adverse Reactions
- Acute Kidney Injury Due to Volume Depletion
- Acute Gallbladder Disease
- Acute Pancreatitis
- Hypersensitivity Reactions
- Hypoglycemia
- Diabetic Retinopathy Complications in Patients with Type 2 Diabetes Mellitus
- Pulmonary Aspiration During General Anesthesia or Deep Sedation
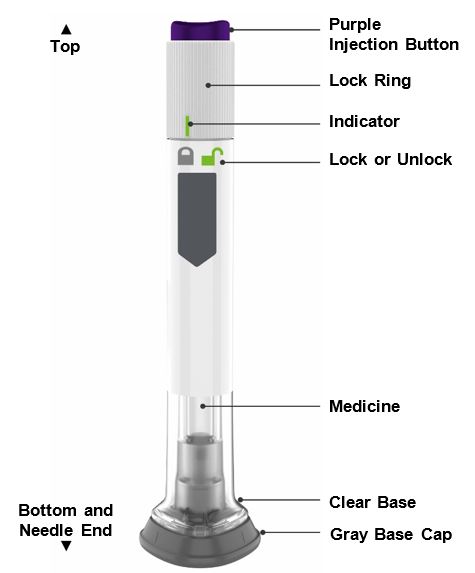
- ZEPBOUND is a single-dose prefilled pen.
- ZEPBOUND is used 1 time each week.
- Inject under the skin (subcutaneously) only.
- You or another person can inject into your stomach (abdomen) or thigh.
- Another person can inject into the back of your upper arm.
- Put your used pen in an FDA-cleared sharps disposal container right away after use.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not recycle your used sharps disposal container.
- Store your pen in the refrigerator between 36°F to 46°F (2°C to 8°C).
- You may store your pen at room temperature up to 86°F (30°C) for up to 21 days. If you store the pen at room temperature, do not return the pen to the refrigerator.
- Discard the pen if not used within 21 days after removing from the refrigerator.
- Do not freeze your pen. If the pen has been frozen, throw the pen away and use a new pen.
- Store your pen in the original carton to protect your pen from light.
- The pen has glass parts. Handle it carefully. If you drop the pen on a hard surface,
- Keep your ZEPBOUND pen and all medicines out of the reach of children.
- If you have vision problems,
- If you have questions or problems with your ZEPBOUND pen, contact Lilly at 1-800-Lilly-Rx (1-800-545-5979) or call your healthcare provider.
- For more information about the ZEPBOUND pen, visit our website at www.zepbound.com.

- ZEPBOUND is a single-dose vial.
- ZEPBOUND is used 1 time each week.
- Inject under the skin (subcutaneously) only.
- You or another person may inject into your stomach (abdomen) or thigh.
- Another person can inject into the back of your upper arm.
- 1 single-dose ZEPBOUND vial
- 1 syringe and 1 needle, supplied separately (for example, use a 1 mL syringe and needle as recommended by your healthcare provider)
- 1 alcohol swab
- gauze
- 1 sharps container for throwing away used needles and syringes.
- Inject exactly as your healthcare provider has shown you. Your healthcare provider should tell you if you should pinch the skin before injecting.
- Change (rotate) your injection site within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not mix ZEPBOUND with any other medicine.
- Do not inject ZEPBOUND in the same injection site used for other medicines.
- Put your used needle and syringe in an FDA-cleared sharps disposal container right away after use.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at:
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Store all unopened vials in the refrigerator at 36°F to 46°F (2°C to 8°C).
- You may store the unopened vial at room temperature up to 86°F (30°C) for up to 21 days.
- Do not freeze. Do not use if ZEPBOUND has been frozen.
- Store the vial in the original carton to protect from light.
- Throw away all opened vials after use, even if there is medicine left in the vial.
- ZEPBOUND is a multi-dose vial.
- Inject
- You or another person may inject into your stomach (abdomen) or thigh.
- Another person can inject into the back of your upper arm.
- 1 multi-dose ZEPBOUND vial
- 1 syringe and 1 needle, supplied separately (for example, use a 1 mL syringe and needle as recommended by your healthcare provider)
- 1 alcohol swab
- gauze
- 1 sharps container for throwing away used needles and syringes.
- Inject exactly as your healthcare provider has shown you. Your healthcare provider should tell you if you should pinch the skin before injecting.
- Change (rotate) your injection site within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not mix ZEPBOUND with any other medicine.
- Do not inject ZEPBOUND in the same injection site used for other medicines.
- Put your used needle and syringe in an FDA-cleared sharps disposal container right away after use.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at:
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Do not freeze. Do not use if ZEPBOUND has been frozen.
- Store ZEPBOUND in original carton. Keep away from light.
- Store unopened vial in the refrigerator at 36°F to 46°F (2°C to 8°C). It can be used until the expiration date on the label if kept in the refrigerator.
- If stored at room temperature [up to 86°F (30°C)], throw away unopened vial after 30 days.
- Store opened (in-use) vial in the original carton in the refrigerator at 36°F to 46°F (2°C to 8°C) or at room temperature [up to 86°F (30°C)]. Throw away opened vial after a total of 30 days at room temperature, 30 days after first use, or after taking 4 weekly doses, even if there is medicine left in it.


- ZEPBOUND KwikPen is a disposable multi-dose single-patient-use prefilled pen.
For your weekly dose, you need to turn the dose knob all the way until it stops AND the - After 4 doses, throw away (discard) the Pen, including the unused medicine. The Pen will prevent you from dialing a full dose after you have given yourself 4 weekly doses.
- Do not share your ZEPBOUND Pen with other people, even if the pen needle has been changed. You may give other people a serious infection or get a serious infection from them.
- People who are blind or have vision problems should not use the Pen without help from a person trained to use the Pen.

- Store
- Unused Pens may be used until the expiration date (EXP) printed on the label if the Pen has been kept in the refrigerator.
- Do not freeze your Pen. Throw away (discard) the Pen if it has been frozen.
- You may store your
- Keep away from heat and light.
- Keep your Pen and needles out of the sight and reach of children.
- Throw away the Pen 30 days after first use even though the Pen has medicine left in it.
- Throw away the Pen after receiving 4 weekly doses. Attempting to inject any leftover medicine could result in an incomplete dose even though the Pen still has medicine left in it.
- Put your used pen needles in an FDA-cleared sharps disposal container right away after use.
- Do not throw away (discard) loose pen needles in your household trash.
- Discard the used Pen in your household trash or FDA-cleared sharps disposal container after you have removed the needle.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away (discard) used needles. For more information about safe sharps disposal, and for specific information about sharps disposal in the state you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- If you cannot remove the Pen cap, gently twist the Pen cap back and forth, and then pull the Pen cap straight off.
- If the dose knob is hard to push:
If the Pen prevents you from turning the dose knob until the - Throw away (discard) the Pen, including the unused medicine. There may not be enough medicine left in the pen to give a full dose.


















![PACKAGE LABEL - Zepbound Multi-Dose Vial - 2.5 mg/0.6 mL Dose (10 mg/2.4 mL [4.17 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-2pt5mg-mdv-787132.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)
![PACKAGE LABEL - Zepbound Multi-Dose Vial - 5 mg/0.6 mL Dose (20 mg/2.4 mL [8.33 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-5mg-mdv-787133.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)
![PACKAGE LABEL - Zepbound Multi-Dose Vial - 7.5 mg/0.6 mL Dose (30 mg/2.4 mL [12.5 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-7pt5mg-mdv-787134.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)
![PACKAGE LABEL - Zepbound Multi-Dose Vial - 10 mg/0.6 mL Dose (40 mg/2.4 mL [16.7 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-10mg-mdv-787135.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)
![PACKAGE LABEL - Zepbound Multi-Dose Vial - 12.5 mg/0.6 mL Dose (50 mg/2.4 mL [20.8 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-12pt5mg-mdv-787136.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)
![PACKAGE LABEL - Zepbound Multi-Dose Vial - 15 mg/0.6 mL Dose (60 mg/2.4 mL [25 mg/mL])](https://dailymed.nlm.nih.gov/dailymed/image.cfm?name=zep-15mg-mdv-787137.jpg&setid=487cd7e7-434c-4925-99fa-aa80b1cc776b)



