Brand Name
Rexulti
Generic Name
Brexpiprazole
View Brand Information FDA approval date: July 10, 2015
Classification: Atypical Antipsychotic
Form: Tablet, Kit
What is Rexulti (Brexpiprazole)?
Brexpiprazole tablets are indicated for: Adjunctive treatment of major depressive disorder in adults. Treatment of schizophrenia in adults. Brexpiprazole tablets is an atypical antipsychotic indicated for: Use as an adjunctive therapy to antidepressants for the treatment of major depressive disorder in adults ( 1, 1.
Approved To Treat
Top Global Experts
There are no experts for this drug
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
There is no clinical trials being done for this treatment
Related Latest Advances
Brand Information
Rexulti (brexpiprazole)
1INDICATIONS AND USAGE
REXULTI is indicated for:
- Adjunctive treatment to antidepressants for major depressive disorder (MDD) in adults
- Treatment of schizophrenia in adults and pediatric patients ages 13 years and older
- Treatment of agitation associated with dementia due to Alzheimer's disease
Limitations of Use:
REXULTI is not indicated as an as needed ("prn") treatment for agitation associated with dementia due to Alzheimer's disease [see
2DOSAGE FORMS AND STRENGTHS
REXULTI tablets are available in 6 strengths:
- 0.25 mg tablets are light brown, round, shallow convex, bevel-edged body with "BRX" and "0.25" imprinted on one side.
- 0.5 mg tablets: are light orange, round, shallow convex, bevel-edged body with "BRX" and "0.5" imprinted on one side.
- 1 mg tablets are light yellow, round, shallow convex, bevel-edged body with "BRX" and "1" imprinted on one side.
- 2 mg tablets are light green, round, shallow convex, bevel-edged body with "BRX" and "2" imprinted on one side.
- 3 mg tablets are light purple, round, shallow convex, bevel-edged body with "BRX" and "3" imprinted on one side.
- 4 mg tablets are white, round, shallow convex, bevel-edged body with "BRX" and "4" imprinted on one side.
3CONTRAINDICATIONS
REXULTI is contraindicated in patients with a known hypersensitivity to brexpiprazole or any of its components. Reactions have included rash, facial swelling, urticaria, and anaphylaxis.
4ADVERSE REACTIONS
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis
- Suicidal Thoughts and Behaviors in Adolescents and Young Adults
- Cerebrovascular Adverse Reactions Including Stroke in Elderly Patients with Dementia-Related Psychosis
- Neuroleptic Malignant Syndrome (NMS)
- Tardive Dyskinesia
- Metabolic Changes
- Pathological Gambling and Other Compulsive Behaviors
- Leukopenia, Neutropenia, and Agranulocytosis
- Orthostatic Hypotension and Syncope
- Falls
- Seizures
- Body Temperature Dysregulation
- Dysphagia
- Potential for Cognitive and Motor Impairment
4.1Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The most common adverse reactions in adult patients in clinical trials (≥5%) were weight increased, akathisia, headache, somnolence, and insomnia.
The most common adverse reactions in pediatric patients in clinical trials (≥5%) were weight increased, somnolence, headache, akathisia, and nasopharyngitis.
Brexpiprazole has been evaluated for safety in 12,550 adult patients who participated in multiple-dose clinical trials for major depressive disorder, schizophrenia, agitation associated with dementia due to Alzheimer's disease, attention deficit hyperactivity disorder (ADHD), post-traumatic stress disorder (PTSD), bipolar mania, and borderline personality disorder (BPD). Among them, 3,870 patients were treated with brexpiprazole for at least 180 days, and 1,910 patients were treated for at least one year of exposure.
Additionally, brexpiprazole has been evaluated for safety in 119 pediatric patients who participated in short-term trials, and 314 patients in long-term multiple-dose clinical trials for pediatric schizophrenia and autism spectrum disorders (ASD).
4.2Postmarketing Experience
The following adverse reaction has been identified during post-approval use of REXULTI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Nervous System disorders: Neuroleptic Malignant Syndrome
5OVERDOSAGE
There is limited clinical trial experience regarding human overdosage with REXULTI.
Management of a REXULTI overdose should concentrate on supportive therapy, maintaining an adequate airway, oxygenation and ventilation, and management of symptoms. Close medical supervision and monitoring should continue until the patient recovers. Consider contacting the Poison Help Line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
Oral activated charcoal and sorbitol (50 g/240 mL), administered one hour after ingesting oral REXULTI, decreased brexpiprazole C
There is no information on the effect of hemodialysis in treating an overdose with REXULTI; hemodialysis is unlikely to be useful because brexpiprazole is highly bound to plasma proteins.
6DESCRIPTION
Brexpiprazole, an atypical antipsychotic, is available as REXULTI
REXULTI tablets are for oral administration and are available in 0.25 mg, 0.5 mg, 1 mg, 2 mg, 3 mg, and 4 mg strengths. Inactive ingredients include lactose monohydrate, corn starch, microcrystalline cellulose, hydroxypropyl cellulose, low-substituted hydroxypropyl cellulose, magnesium stearate, hypromellose, and talc. Colorants include titanium dioxide, iron oxide, and ferrosoferric oxide.
7PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide).
8PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Label
0.25 mg
30 Tablets
NDC 59148-035-13
NDC 59148-035-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck

9PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Carton
0.25 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
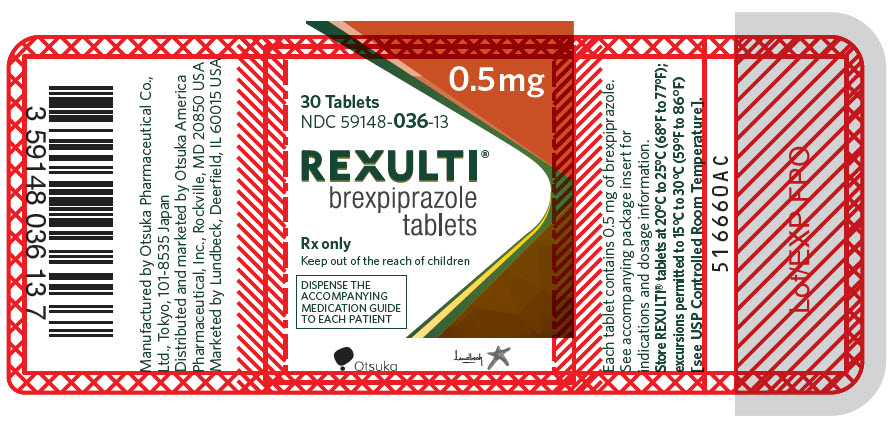
10PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Label
0.5 mg
30 Tablets
NDC 59148-036-13
NDC 59148-036-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck

11PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Carton
0.5 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
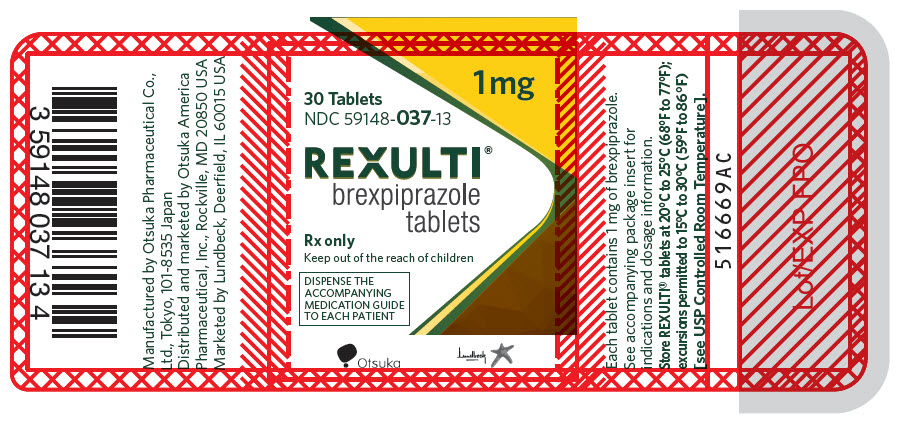
12PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label
1 mg
30 Tablets
NDC 59148-037-13
NDC 59148-037-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck
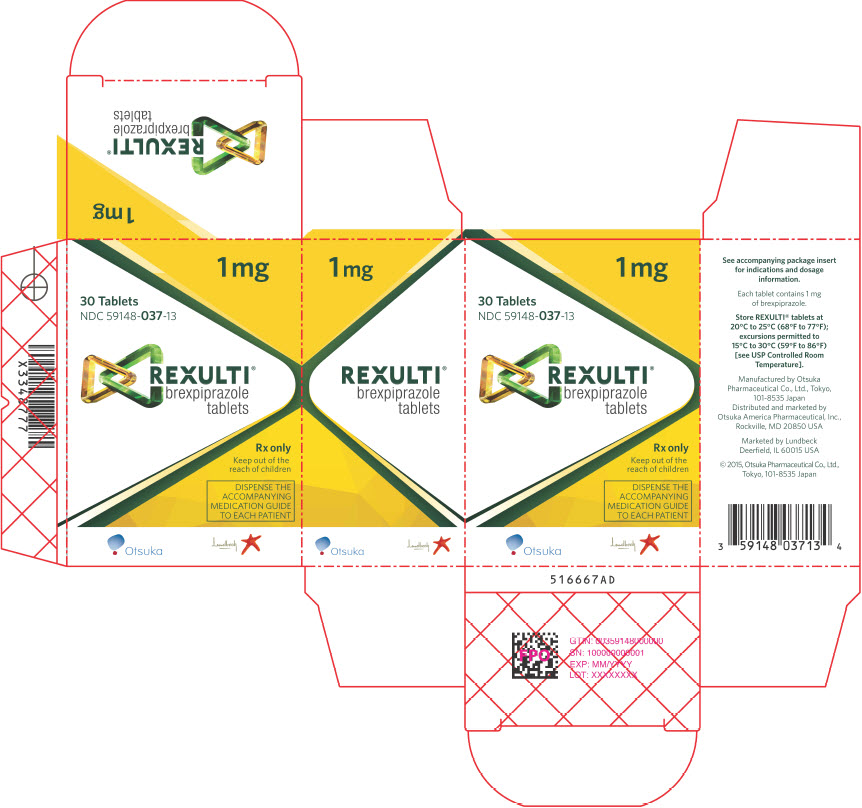
13PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Carton
1 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
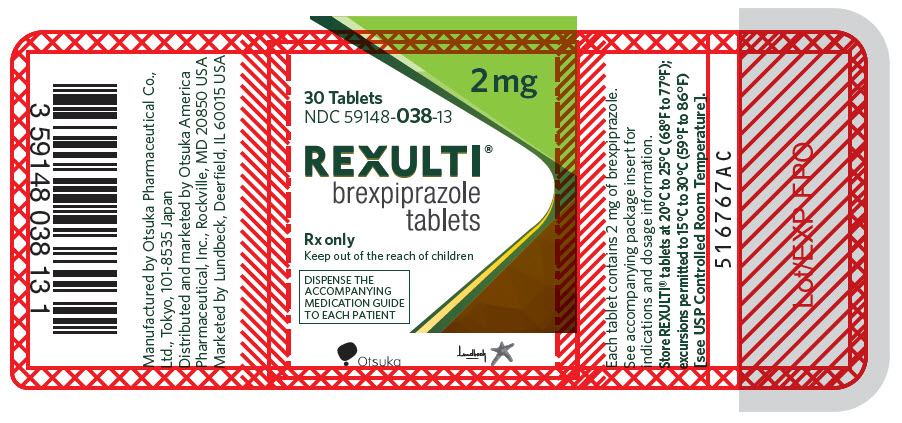
14PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Label
2 mg
30 Tablets
NDC 59148-038-13
NDC 59148-038-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck

15PRINCIPAL DISPLAY PANEL - 2 mg Tablet Bottle Carton
2 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
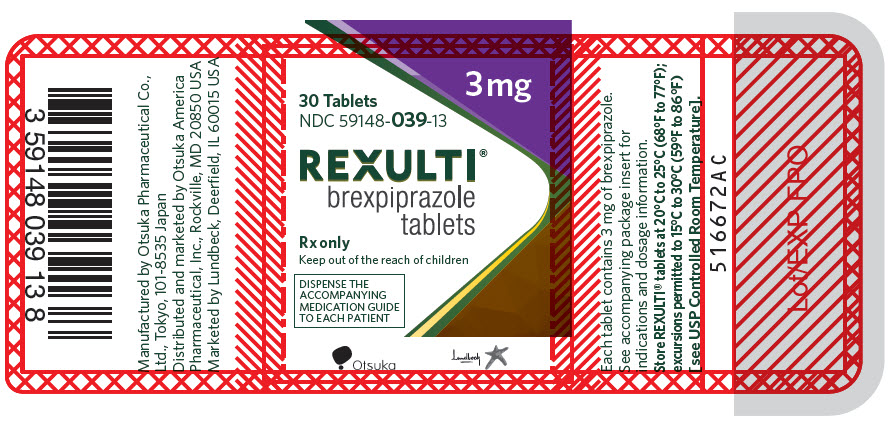
16PRINCIPAL DISPLAY PANEL - 3 mg Tablet Bottle Label
3 mg
30 Tablets
NDC 59148-039-13
NDC 59148-039-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck
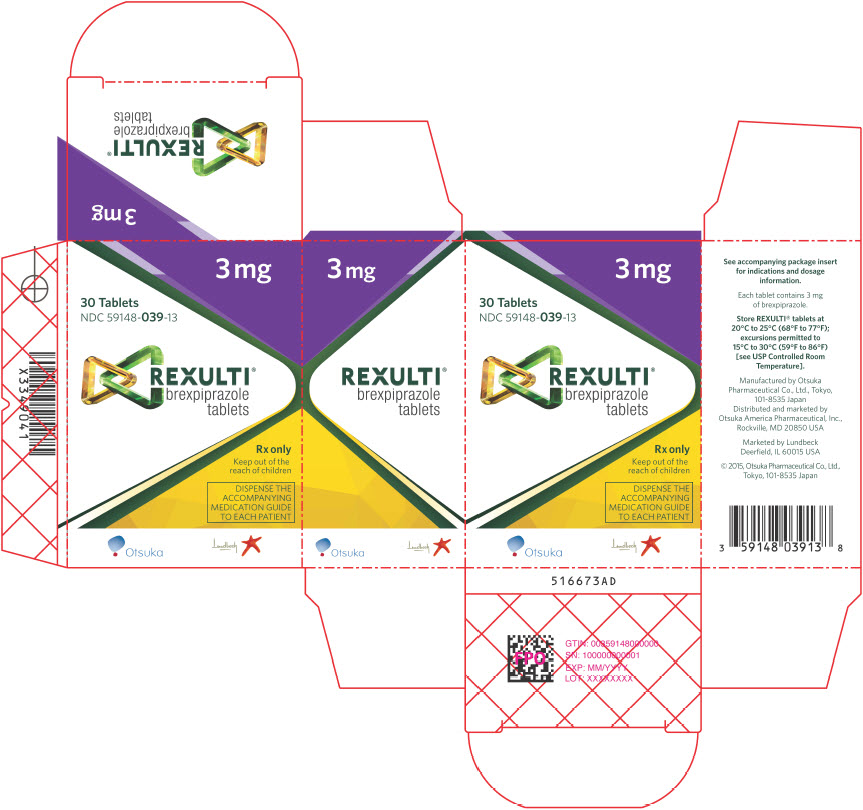
17PRINCIPAL DISPLAY PANEL - 3 mg Tablet Bottle Carton
3 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
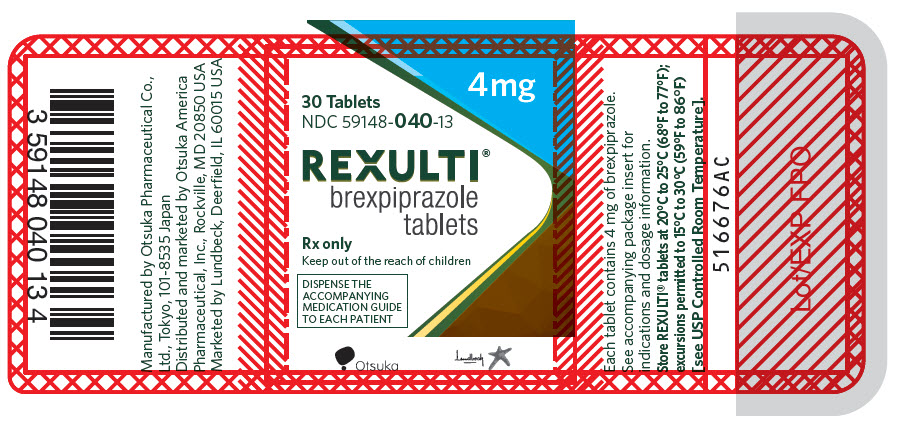
18PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Label
4 mg
30 Tablets
NDC 59148-040-13
NDC 59148-040-13
REXULTI
Rx only
Keep out of the reach of children
DISPENSE THE
Otsuka
Lundbeck
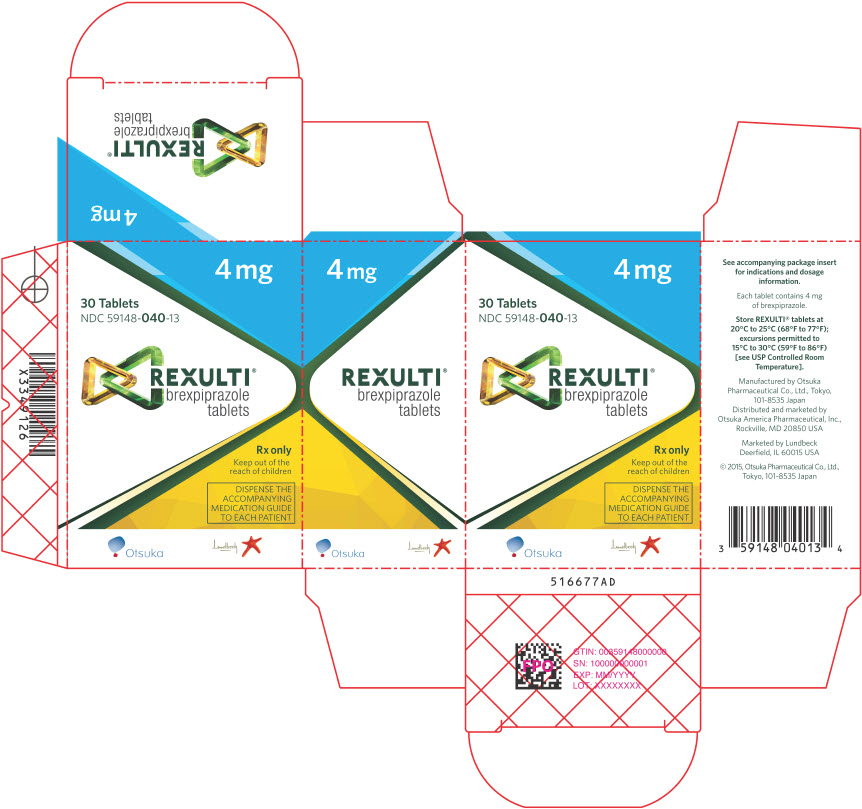
19PRINCIPAL DISPLAY PANEL - 4 mg Tablet Bottle Carton
4 mg
30 Tablets
REXULTI
Rx only
Keep out of the
DISPENSE THE
