Brand Name
OXAliplatin
View Brand InformationFDA approval date: August 10, 2009
Classification: Platinum-based Drug
Form: Injection
What is OXAliplatin?
Oxaliplatin injection, in combination with infusional fluorouracil and leucovorin, is indicated for: adjuvant treatment of stage III colon cancer in patients who have undergone complete resection of the primary tumor., treatment of advanced colorectal cancer. Oxaliplatin injection is a platinum-based drug used in combination with infusional fluorouracil and leucovorin, which is indicated for:, adjuvant treatment of stage III colon cancer in patients who have undergone complete resection of the primary tumor. , treatment of advanced colorectal cancer.
Approved To Treat
Top Global Experts
There are no experts for this drug
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
There is no clinical trials being done for this treatment
Related Latest Advances
There is no latest advances for this treatment
Brand Information
Oxaliplatin (Oxaliplatin)
WARNING: ANAPHYLACTIC REACTIONS
Anaphylactic reactions to oxaliplatin for injection have been reported, and may occur within minutes of oxaliplatin for injection administration. Epinephrine, corticosteroids, and antihistamines have been employed to alleviate symptoms of anaphylaxis
1INDICATIONS & USAGE
Oxaliplatin for injection USP, used in combination with infusional 5-fluorouracil/leucovorin, is indicated for:
2DOSAGE & ADMINISTRATION
Oxaliplatin for injection should be administered under the supervision of a qualified physician experienced in the use of cancer chemotherapeutic agents. Appropriate management of therapy and complications is possible only when adequate diagnostic and treatment facilities are readily available.
2.1Dosage
Administer oxaliplatin for injection in combination with 5-fluorouracil/leucovorin every 2 weeks. For advanced disease, treatment is recommended until disease progression or unacceptable toxicity. For adjuvant use, treatment is recommended for a total of 6 months (12 cycles):
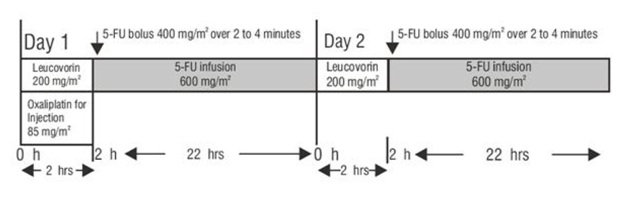
The administration of oxaliplatin for injection does not require prehydration. Premedication with antiemetics, including 5-HT
2.2Dose Modification Recommendations
Prior to subsequent therapy cycles, patients should be evaluated for clinical toxicities and recommended laboratory tests
2.3Preparation of Infusion Solution
Reconstitution or final dilution must never be performed with a sodium chloride solution or other chloride containing solutions.The lyophilized powder is reconstituted by adding 10 mL (for the 50 mg vial) or 20 mL (for the 100 mg vial) of Water for Injection, USP or 5% Dextrose Injection, USP. Do not administer the reconstituted solution without further dilution. The reconstituted solution must be further diluted in an infusion solution of 250 to 500 mL of 5% Dextrose Injection, USP.
After reconstitution in the original vial, the solution may be stored up to 24 hours under refrigeration [2° to 8°C (36° to 46°F)]. After final dilution with 250 to 500 mL of 5% Dextrose Injection, USP, the shelf life is 6 hours at room temperature [20° to 25°C (68° to 77°F)] or up to 24 hours under refrigeration [2° to 8°C (36° to 46°F)].Oxaliplatin for injection is not light sensitive.
Oxaliplatin for injection is incompatible in solution with alkaline medications or media such as basic solutions of 5-fluorouracil) and must not be mixed with these or administered simultaneously through the same infusion line. The infusion line should be flushed with 5% Dextrose Injection, USP prior to administration of any concomitant medication.Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration and discarded if present.
Needles or intravenous administration sets containing aluminum parts that may come in contact with oxaliplatin for injection should not be used for the preparation or mixing of the drug. Aluminum has been reported to cause degradation of platinum compounds.
After reconstitution in the original vial, the solution may be stored up to 24 hours under refrigeration [2° to 8°C (36° to 46°F)]. After final dilution with 250 to 500 mL of 5% Dextrose Injection, USP, the shelf life is 6 hours at room temperature [20° to 25°C (68° to 77°F)] or up to 24 hours under refrigeration [2° to 8°C (36° to 46°F)].Oxaliplatin for injection is not light sensitive.
Oxaliplatin for injection is incompatible in solution with alkaline medications or media such as basic solutions of 5-fluorouracil) and must not be mixed with these or administered simultaneously through the same infusion line. The infusion line should be flushed with 5% Dextrose Injection, USP prior to administration of any concomitant medication.Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration and discarded if present.
Needles or intravenous administration sets containing aluminum parts that may come in contact with oxaliplatin for injection should not be used for the preparation or mixing of the drug. Aluminum has been reported to cause degradation of platinum compounds.
3DOSAGE FORMS & STRENGTHS
Oxaliplatin for injection is supplied in single-dose vials containing 50 mg or 100 mg of oxaliplatin as a sterile, preservative-free lyophilized powder for reconstitution.
4CONTRAINDICATIONS
Oxaliplatin for injection should not be administered to patients with a history of known allergy to oxaliplatin for injection or other platinum compounds
5ADVERSE REACTIONS
The following serious adverse reactions are discussed in greaterdetail in other sections of the label:
- Anaphylaxis and Allergic reactions
- Neuropathy
- Severe Neutropenia
- Pulmonary Toxicities
- Hepatotoxicity
- Cardiovascular Toxicities
- Rhabdomyolysis
5.1Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
1Includes thrombosis related to the catheter
The following table provides adverse reactions reported in the adjuvant therapy colon cancer clinical trial [see Clinical Studies (14)] by body system and decreasing order of frequency in the oxaliplatin for injection and infusional 5-fluorouracil/leucovorin arm for events with overall incidences ≥ 5% but with incidences <1% NCI grade 3/4 events.
Table 4 - Adverse Reactions Reported in Patients with Colon Cancer receiving Adjuvant Treatment (≥ 5% of all patients, but with <1% NCI Grade 3/4 events)
The following table provides adverse reactions reported in the adjuvant therapy colon cancer clinical trial [see Clinical Studies (14)] by body system and decreasing order of frequency in the oxaliplatin for injection and infusional 5-fluorouracil/leucovorin arm for events with overall incidences ≥ 5% but with incidences <1% NCI grade 3/4 events.
Table 4 - Adverse Reactions Reported in Patients with Colon Cancer receiving Adjuvant Treatment (≥ 5% of all patients, but with <1% NCI Grade 3/4 events)
Although specific events can vary, the overall frequency of adverse reactions was similar in men and women and in patients <65 and ≥65 years. However, the following grade 3/4 events were more common in females: diarrhea, fatigue, granulocytopenia, nausea and vomiting. In patients ≥65 years old, the incidence of grade 3/4 diarrhea and granulocytopenia was higher than in younger patients. Insufficient subgroup sizes prevented analysis of safety by race. The following additional adverse reactions, were reported in ≥2% and <5% of the patients in the oxaliplatin for injection and infusional 5-fluorouracil/leucovorin combination arm (listed in decreasing order of frequency): pain, leukopenia, weight decrease, coughing.
* Not otherwise specified
* Absolute neutrophil count
The following table provides adverse reactions reported in the previously treated study
Adverse reactions were similar in men and women and in patients <65 and ≥65 years, but older patients may have been more susceptible to dehydration, diarrhea, hypokalemia and fatigue. The following additional adverse reactions, at least possibly related to treatment and potentially important, were reported in ≥2% and <5% of the patients in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm (listed in decreasing order of frequency): anxiety, myalgia, erythematous rash, increased sweating, conjunctivitis, weight decrease, dry mouth, rectal hemorrhage, depression, ataxia, ascites, hemorrhoids, muscle weakness, nervousness, tachycardia, abnormal micturition frequency, dry skin, pruritus, hemoptysis, purpura, vaginal hemorrhage, melena, somnolence, pneumonia, proctitis, involuntary muscle contractions, intestinal obstruction, gingivitis, tenesmus, hot flashes, enlarged abdomen, urinary incontinence.
Table 10 – Adverse Hematologic Reactions in Patients Previously Untreated for Advanced Colorectal Cancer (≥5% of patients)
Table 11 – Adverse Hematologic Reactions in Previously Treated Patients (≥5% of patients)
Thrombocytopenia and BleedingThrombocytopenia was frequently reported with the combination of oxaliplatin for injection and infusional 5-fluorouracil/leucovorin. The incidence of all hemorrhagic events in the adjuvant and previously treated patients was higher on the oxaliplatin for injection combination arm compared to the infusional 5-fluorouracil/leucovorin arm. These events included gastrointestinal bleeding, hematuria, and epistaxis. In the adjuvant trial, two patients died from intracerebral hemorrhages.
The incidence of Grade 3/4 thrombocytopenia was 2% in adjuvant patients with colon cancer. In patients treated for advanced colorectal cancer the incidence of Grade 3/4 thrombocytopenia was 3 to 5%, and the incidence of these events was greater for the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin over the irinotecan plus 5-fluorouracil/leucovorin or 5-fluorouracil/leucovorin control groups. Grade 3/4 gastrointestinal bleeding was reported in 0.2% of adjuvant patients receiving oxaliplatin for injection and 5-fluorouracil/leucovorin. In the previously untreated patients, the incidence of epistaxis was 10% in the oxaliplatin for injection and 5-fluorouracil/leucovorin arm, and 2% and 1%, respectively, in the irinotecan plus 5-fluorouracil/leucovorin or irinotecan plus oxaliplatin for injection arms.
Neutropenia
Neutropenia was frequently observed with the combination of oxaliplatin for injection and 5-
fluorouracil/leucovorin, with Grade 3 and 4 events reported in 29% and 12% of adjuvant patients with colon cancer, respectively. In the adjuvant trial, 3 patients died from sepsis/neutropenic sepsis. Grade 3 and 4 events were reported in 35% and 18% of the patients previously untreated for advanced colorectal cancer, respectively. Grade 3 and 4 events were reported in 27% and 17% of previously treated patients, respectively. In adjuvant patients the incidence of either febrile neutropenia (0.7%) or documented infection with concomitant grade 3/4 neutropenia (1.1%) was 1.8% in the oxaliplatin for injection and 5-fluorouracil/leucovorin arm. The incidence of febrile neutropenia in the patients previously untreated for advanced colorectal cancer was 15% (3% of cycles) in the irinotecan plus 5-fluorouracil/leucovorin arm and 4% (less than 1% of cycles) in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm. Additionally, in this same population, infection with grade 3 or 4 neutropenia was 12% in the irinotecan plus 5-fluorouracil/leucovorin, and 8% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination. The incidence of febrile neutropenia in the previously treated patients was 1% in the 5-fluorouracil/leucovorin arm and 6% (less than 1% of cycles) in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm.
Gastrointestinal
In patients receiving the combination of oxaliplatin for injection plus infusional 5-fluorouracil/leucovorin for adjuvant treatment for colon cancer the incidence of Grade 3/4 nausea and vomiting was greater than those receiving infusional 5-fluorouracil/leucovorin alone (see table). In patients previously untreated for advanced colorectal cancer receiving the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin, the incidence of Grade 3 and 4 vomiting and diarrhea was less compared to irinotecan plus 5-fluorouracil/leucovorin controls (see table). In previously treated patients receiving the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin, the incidence of Grade 3 and 4 nausea, vomiting, diarrhea, and mucositis/stomatitis increased compared to 5-fluorouracil/leucovorin controls (see table).
The incidence of gastrointestinal adverse reactions in the previously untreated and previously treated patients appears to be similar across cycles. Premedication with antiemetics, including 5-HT3 blockers, is recommended. Diarrhea and mucositis may be exacerbated by the addition of oxaliplatin for injection to 5-fluorouracil/leucovorin, and should be managed with appropriate supportive care. Since cold temperature can exacerbate acute neurological symptoms, ice (mucositis prophylaxis) should be avoided during the infusion of oxaliplatin for injection.
Dermatologic
Oxaliplatin for injection did not increase the incidence of alopecia compared to 5-fluorouracil/leucovorin alone. No complete alopecia was reported. The incidence of Grade 3/4 skin disorders was 2% in both the oxaliplatin for injection plus infusional 5-fluorouracil/leucovorin and the infusional 5-fluorouracil/leucovorin alone arms in the adjuvant colon cancer patients. The incidence of hand-foot syndrome in patients previously untreated for advanced colorectal cancer was 2% in the irinotecan plus 5-fluorouracil/leucovorin arm and 7% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm. The incidence of hand-foot syndrome in previously treated patients was 13% in the 5-fluorouracil/leucovorin arm and 11% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm.
Intravenous Site ReactionsExtravasation, in some cases including necrosis, has been reported.
Injection site reaction, including redness, swelling, and pain, has been reported.
Anticoagulation and HemorrhageThere have been reports while on study and from post-marketing surveillance of prolonged prothrombin time and INR occasionally associated with hemorrhage in patients who received oxaliplatin for injection plus 5-fluorouracil/leucovorin while on anticoagulants. Patients receiving oxaliplatin for injection plus 5-fluorouracil/leucovorin and requiring oral anticoagulants may require closer monitoring.
RenalAbout 5 to 10% of patients in all groups had some degree of elevation of serum creatinine. The incidence of Grade 3/4 elevations in serum creatinine in the oxaliplatin for injection and 5- fluorouracil/leucovorin combination arm was 1% in the previously treated patients. Serum creatinine measurements were not reported in the adjuvant trial.
Hepatic
Hepatotoxicity (defined as elevation of liver enzymes) appears to be related to oxaliplatin for injection combination therapy [see Warnings and Precautions (5.6)]. The following tables list the clinical chemistry changes associated with hepatic toxicity occurring in ≥5% of patients, based on adverse reactions reported and NCI CTC grade for adjuvant patients and patients previously untreated for advanced colorectal cancer, laboratory values and NCI CTC grade for previously treated patients.
Table 12 - Adverse Hepatic Reactions in Patients with Stage II or III Colon Cancer Receiving Adjuvant Therapy (≥5% of patients)
The incidence of Grade 3/4 thrombocytopenia was 2% in adjuvant patients with colon cancer. In patients treated for advanced colorectal cancer the incidence of Grade 3/4 thrombocytopenia was 3 to 5%, and the incidence of these events was greater for the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin over the irinotecan plus 5-fluorouracil/leucovorin or 5-fluorouracil/leucovorin control groups. Grade 3/4 gastrointestinal bleeding was reported in 0.2% of adjuvant patients receiving oxaliplatin for injection and 5-fluorouracil/leucovorin. In the previously untreated patients, the incidence of epistaxis was 10% in the oxaliplatin for injection and 5-fluorouracil/leucovorin arm, and 2% and 1%, respectively, in the irinotecan plus 5-fluorouracil/leucovorin or irinotecan plus oxaliplatin for injection arms.
Neutropenia
Neutropenia was frequently observed with the combination of oxaliplatin for injection and 5-
fluorouracil/leucovorin, with Grade 3 and 4 events reported in 29% and 12% of adjuvant patients with colon cancer, respectively. In the adjuvant trial, 3 patients died from sepsis/neutropenic sepsis. Grade 3 and 4 events were reported in 35% and 18% of the patients previously untreated for advanced colorectal cancer, respectively. Grade 3 and 4 events were reported in 27% and 17% of previously treated patients, respectively. In adjuvant patients the incidence of either febrile neutropenia (0.7%) or documented infection with concomitant grade 3/4 neutropenia (1.1%) was 1.8% in the oxaliplatin for injection and 5-fluorouracil/leucovorin arm. The incidence of febrile neutropenia in the patients previously untreated for advanced colorectal cancer was 15% (3% of cycles) in the irinotecan plus 5-fluorouracil/leucovorin arm and 4% (less than 1% of cycles) in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm. Additionally, in this same population, infection with grade 3 or 4 neutropenia was 12% in the irinotecan plus 5-fluorouracil/leucovorin, and 8% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination. The incidence of febrile neutropenia in the previously treated patients was 1% in the 5-fluorouracil/leucovorin arm and 6% (less than 1% of cycles) in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm.
Gastrointestinal
In patients receiving the combination of oxaliplatin for injection plus infusional 5-fluorouracil/leucovorin for adjuvant treatment for colon cancer the incidence of Grade 3/4 nausea and vomiting was greater than those receiving infusional 5-fluorouracil/leucovorin alone (see table). In patients previously untreated for advanced colorectal cancer receiving the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin, the incidence of Grade 3 and 4 vomiting and diarrhea was less compared to irinotecan plus 5-fluorouracil/leucovorin controls (see table). In previously treated patients receiving the combination of oxaliplatin for injection and 5-fluorouracil/leucovorin, the incidence of Grade 3 and 4 nausea, vomiting, diarrhea, and mucositis/stomatitis increased compared to 5-fluorouracil/leucovorin controls (see table).
The incidence of gastrointestinal adverse reactions in the previously untreated and previously treated patients appears to be similar across cycles. Premedication with antiemetics, including 5-HT3 blockers, is recommended. Diarrhea and mucositis may be exacerbated by the addition of oxaliplatin for injection to 5-fluorouracil/leucovorin, and should be managed with appropriate supportive care. Since cold temperature can exacerbate acute neurological symptoms, ice (mucositis prophylaxis) should be avoided during the infusion of oxaliplatin for injection.
Dermatologic
Oxaliplatin for injection did not increase the incidence of alopecia compared to 5-fluorouracil/leucovorin alone. No complete alopecia was reported. The incidence of Grade 3/4 skin disorders was 2% in both the oxaliplatin for injection plus infusional 5-fluorouracil/leucovorin and the infusional 5-fluorouracil/leucovorin alone arms in the adjuvant colon cancer patients. The incidence of hand-foot syndrome in patients previously untreated for advanced colorectal cancer was 2% in the irinotecan plus 5-fluorouracil/leucovorin arm and 7% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm. The incidence of hand-foot syndrome in previously treated patients was 13% in the 5-fluorouracil/leucovorin arm and 11% in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm.
Intravenous Site ReactionsExtravasation, in some cases including necrosis, has been reported.
Injection site reaction, including redness, swelling, and pain, has been reported.
Anticoagulation and HemorrhageThere have been reports while on study and from post-marketing surveillance of prolonged prothrombin time and INR occasionally associated with hemorrhage in patients who received oxaliplatin for injection plus 5-fluorouracil/leucovorin while on anticoagulants. Patients receiving oxaliplatin for injection plus 5-fluorouracil/leucovorin and requiring oral anticoagulants may require closer monitoring.
RenalAbout 5 to 10% of patients in all groups had some degree of elevation of serum creatinine. The incidence of Grade 3/4 elevations in serum creatinine in the oxaliplatin for injection and 5- fluorouracil/leucovorin combination arm was 1% in the previously treated patients. Serum creatinine measurements were not reported in the adjuvant trial.
Hepatic
Hepatotoxicity (defined as elevation of liver enzymes) appears to be related to oxaliplatin for injection combination therapy [see Warnings and Precautions (5.6)]. The following tables list the clinical chemistry changes associated with hepatic toxicity occurring in ≥5% of patients, based on adverse reactions reported and NCI CTC grade for adjuvant patients and patients previously untreated for advanced colorectal cancer, laboratory values and NCI CTC grade for previously treated patients.
Table 12 - Adverse Hepatic Reactions in Patients with Stage II or III Colon Cancer Receiving Adjuvant Therapy (≥5% of patients)
Table 13 – Adverse Hepatic Clinical Chemistry Abnormalities in Patients Previously Untreated for Advanced Colorectal Cancer (≥5% of patients)
Table 14 – Adverse Hepatic Clinical Chemistry Abnormalities in Previously Treated Patients (≥5% of patients)
Thromboembolism
The incidence of thromboembolic events in adjuvant patients with colon cancer was 6% (1.8% grade 3/4) in the infusional 5-fluorouracil/leucovorin arm and 6% (1.2% grade 3/4) in the oxaliplatin for injection and infusional 5-fluorouracil/leucovorin combined arm, respectively. The incidence was 6 and 9% of the patients previously untreated for advanced colorectal cancer and previously treated patients in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm, respectively.
The incidence of thromboembolic events in adjuvant patients with colon cancer was 6% (1.8% grade 3/4) in the infusional 5-fluorouracil/leucovorin arm and 6% (1.2% grade 3/4) in the oxaliplatin for injection and infusional 5-fluorouracil/leucovorin combined arm, respectively. The incidence was 6 and 9% of the patients previously untreated for advanced colorectal cancer and previously treated patients in the oxaliplatin for injection and 5-fluorouracil/leucovorin combination arm, respectively.
5.2Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oxaliplatin for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a whole:
angioedema, anaphylactic shock
Cardiovascular disorders:
QT prolongation leading to ventricular arrhythmias including fatal Torsade de Pointes
Central and peripheral nervous system disorders:
loss of deep tendon reflexes, dysarthria, Lhermitte’s sign, cranial nerve palsies, fasciculations, convulsion, Reversible Posterior Leukoencephalopathy Syndrome (RPLS, also known as PRES).
Hearing and vestibular system disorders:
deafness
Infections:
septic shock, including fatal outcomes
Infusion reactions/hypersensitivity:
laryngospasm
Liver and Gastrointestinal system disorders:
severe diarrhea/vomiting resulting in hypokalemia, colitis (including
Musculoskeletal and connective tissue disorders
rhabdomyolysis, including fatal outcomes.
Platelet, bleeding, and clotting disorders: immuno-allergic thrombocytopenia prolongation of prothrombin time and of INR in patients receiving anticoagulants
Red Blood Cell disorders:
hemolytic uremic syndrome, immuno-allergic hemolytic anemia
Renal disorders:
Acute tubular necrosis, acute interstitial nephritis and acute renal failure.
Respiratory system disorders:
pulmonary fibrosis, and other interstitial lung diseases (sometimes fatal)
Vision disorders:
decrease of visual acuity, visual field disturbance, optic neuritis and transient vision loss
(reversible following therapy discontinuation)
6DRUG INTERACTIONS
No specific cytochrome P-450-based drug interaction studies have been conducted. No pharmacokinetic interaction between 85 mg/m2 oxaliplatin for injection and 5-fluorouracil/leucovorin has been observed in patients treated every 2 weeks. Increases of 5-fluorouracil plasma concentrations by approximately 20% have been observed with doses of 130 mg/m2 oxaliplatin for injection dosed every 3 weeks. Because platinum-containing species are eliminated primarily through the kidney, clearance of these products may be decreased by coadministration of potentially nephrotoxic compounds; although, this has not been specifically studied [
7OVERDOSAGE
There is no known antidote for oxaliplatin for injection overdose. In addition to thrombocytopenia, the anticipated complications of an oxaliplatin for injection overdose include hypersensitivity reaction, myelosuppression, nausea, vomiting, diarrhea and neurotoxicity.
Several cases of overdoses have been reported with oxaliplatin for injection. Adverse reactions observed were Grade 4 thrombocytopenia (<25,000/mm
Patients suspected of receiving an overdose should be monitored, and supportive treatment should be administered. The maximum dose of oxaliplatin that has been administered in a single infusion is 825 mg.
8DESCRIPTION
Oxaliplatin for injection USP is an antineoplastic agent with the molecular formula C
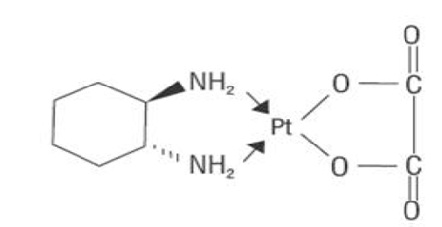
The molecular weight is 397.3. Oxaliplatin is slightly soluble in water at 6 mg/mL, very slightly soluble in methanol, and practically insoluble in ethanol and acetone.
9REFERENCES
1. NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004-165.
10PATIENT COUNSELING INFORMATION
Advise patients:
- To expect side effects of oxaliplatin for injection, particularly its neurologic effects, both the acute, reversible effects and the persistent neurosensory toxicity. Patients should be informed that the acute neurosensory toxicity may be precipitated or exacerbated by exposure to cold or cold objects.
- To avoid cold drinks, use of ice, and should cover exposed skin prior to exposure to cold temperature or cold objects.
- Of the risk of low blood cell counts and to contact their physician immediately should fever, particularly if associated with persistent diarrhea, or evidence of infection develop.
- To contact their physician if persistent vomiting, diarrhea, signs of dehydration, cough or breathing difficulties occur, or signs of allergic reaction appear.
- To exercise caution when driving and using machines. No studies on the effects of the ability to operate cars and machines have been performed; however, oxaliplatin treatment resulting in an increase risk of dizziness, nausea and vomiting, and other neurologic symptoms that affect gait and balance may lead to a minor or moderate influence on the ability to drive and use machines.
- Of the potential effects of vision abnormalities, in particular transient vision loss (reversible following therapy discontinuation), which may affect patients' ability to drive and use machines.
FDA-Approved Patient Labeling
Manufactured by:
11PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
OXALIPLATIN FOR INJECTION, USP
Vial Label
Carton Label
OXALIPLATIN FOR INJECTION, USP
100 mg/Vial
Cytotoxic Agent
FOR INTRAVENOUS USE ONLY
SINGLE DOSE VIAL
Lyophilized
Must be reconstituted and diluted before use
Vial Label
Carton Label
Vial Label
Carton Label
OXALIPLATIN FOR INJECTION, USP
100 mg/Vial
Cytotoxic Agent
FOR INTRAVENOUS USE ONLY
SINGLE DOSE VIAL
Lyophilized
Must be reconstituted and diluted before use
Vial Label
Carton Label