Brand Name
Teflaro
Generic Name
Ceftaroline Fosamil
View Brand Information FDA approval date: October 29, 2010
Classification: Cephalosporin Antibacterial
Form: Injection, Powder
What is Teflaro (Ceftaroline Fosamil)?
Ceftaroline fosamil for injection is a cephalosporin antibacterial indicated in adult and pediatric patients for the treatment of the following infection caused by designated susceptible bacteria: Acute bacterial skin and skin structure infections in adult and pediatric patients .
Approved To Treat
Save this treatment for later
Not sure about your diagnosis?
Related Clinical Trials
There is no clinical trials being done for this treatment
Related Latest Advances
Brand Information
Teflaro (ceftaroline fosamil)
1DOSAGE FORMS AND STRENGTHS
For Injection: Teflaro is supplied as 600 mg or 400 mg of pale yellowish-white to light yellow sterile ceftaroline fosamil (equivalent to 668 mg and 446 mg, respectively, of ceftaroline fosamil monoacetate monohydrate) powder in single-dose, 20 mL clear glass vials. The powder is constituted and further diluted for intravenous injection.
2CONTRAINDICATIONS
Teflaro is contraindicated in patients with known serious hypersensitivity to ceftaroline or other members of the cephalosporin class. Anaphylaxis has been reported with ceftaroline.
3ADVERSE REACTIONS
The following serious adverse reactions are described in greater detail in the Warnings and Precautions section
- Hypersensitivity Reactions
- Clostridioidesdifficile-Associated diarrhea [seeWarnings and Precautions(5.2)]
- Neurological Adverse Reactions
- Direct Coombs’ Test Seroconversion
3.1Clinical TrialsExperience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be compared directly to rates from clinical trials of another drug and may not reflect rates observed in practice.
Adult Patients
Teflaro was evaluated in four controlled comparative Phase 3 clinical trials (two in ABSSSI and two in CABP) which included 1300 adult patients treated with Teflaro (600 mg administered by IV over 1 hour every 12h) and 1297 patients treated with comparator (vancomycin plus aztreonam or ceftriaxone) for a treatment period up to 21 days. The median age of patients treated with Teflaro was 54 years, ranging between 18 and 99 years old. Patients treated with Teflaro were predominantly male (63%) and Caucasian (82%).
Serious Adverse Reactions and Adverse Reactions Leading to Discontinuation
In the four pooled adult Phase 3 clinical trials, serious adverse reactions (SARs) occurred in 98/1300 (7.5%) of patients receiving Teflaro and 100/1297 (7.7%) of patients receiving comparator drugs. Treatment discontinuation due to adverse reactions occurred in 35/1300 (2.7%) of patients receiving Teflaro and 48/1297 (3.7%) of patients receiving comparator drugs with the most common adverse reactions leading to discontinuation being hypersensitivity for both treatment groups at a rate of 0.3% in the Teflaro group and 0.5% in comparator group.
Most Common Adverse Reactions
No adverse reactions occurred in greater than 5% of adult patients receiving Teflaro. The most common adverse reactions occurring in > 2% of patients receiving Teflaro in the pooled adult phase 3 clinical trials were diarrhea, nausea, and rash.
Table 6 lists adverse reactions occurring in ≥ 2% of patients receiving Teflaro in the pooled adult Phase 3 clinical trials.
a Comparators included vancomycin 1 gram IV every 12h plus aztreonam 1 gram IV every 12h in the Phase 3 ABSSSI trials, and ceftriaxone 1 gram IV every 24h in the Phase 3 CABP trials.
Other Adverse Reactions Observed During Clinical Trials of Teflaro
Following is a list of additional adverse reactions reported by the 1740 adult patients who received Teflaro in any clinical trial with incidences less than 2%.
Blood and lymphatic system disorders - Anemia, Eosinophilia, Neutropenia, Thrombocytopenia
Cardiac disorders - Bradycardia, Palpitations
Gastrointestinal disorders - Abdominal pain
General disorders and administration site conditions - Pyrexia
Hepatobiliary disorders - Hepatitis
Immune system disorders - Hypersensitivity, Anaphylaxis
Infections and infestations - Clostridioides difficile colitis
Metabolism and nutrition disorders - Hyperglycemia, Hyperkalemia
Nervous system disorders - Dizziness, Convulsion
Renal and urinary disorders - Renal failure
Skin and subcutaneous tissue disorders - Urticaria
Pediatric Patients
Teflaro was evaluated in three clinical trials (one in ABSSSI and two in CABP) which included 257 pediatric patients 2 months to < 18 years of age treated with Teflaro, and 102 patients treated with comparator agents for a treatment period up to 21 days. In two trials, one in ABSSSI and one in CABP, the dose was selected to result in exposures comparable to adult exposure with 600 mg administered by IV infusion every 12h. In an additional pediatric trial in complicated CABP the dose was higher. The median age of pediatric patients treated with Teflaro was 5 years, ranging from 2 months to < 18 years of age. Patients treated with Teflaro were predominantly male (55%) and Caucasian (92%).
A single study enrolled 11 pediatric patients with a gestational age of ≥34 weeks and a postnatal age of 12 days to less than 2 months of age. The safety findings were similar to those observed in adult and pediatric patients 2 months of age and older.
Serious Adverse Reactions and Adverse Reactions Leading to Discontinuation
In the three pooled pediatric clinical trials, SARs occurred in 10/257 (4%) of patients receiving Teflaro and 3/102 (3%) of patients receiving comparator drugs. Treatment discontinuation due to adverse reactions occurred in 10/257 (3.9%) of patients receiving Teflaro and 2/102 (2%) of patients receiving comparator drugs with the most common adverse reaction leading to discontinuation being rash in 2/257 (0.8%) of patients treated with Teflaro.
Most Common Adverse Reactions
No adverse reactions occurred in greater than 8% of pediatric patients receiving Teflaro. The most common adverse reactions occurring in
Table 7 lists adverse reactions occurring in ≥ 3% of patients receiving Teflaro in the pooled pediatric clinical trials.
a Comparators included vancomycin or cefazolin with or without aztreonam in the ABSSSI trial and ceftriaxone alone or ceftriaxone plus vancomycin in the CABP trials
Following is a list of additional adverse reactions reported by the 257 patients who received Teflaro in the pediatric clinical trials with incidences less than 3%.
Investigations – Alanine aminotransferase increased, Aspartate aminotransferase increased
Nervous system disorders – Headache
Skin and subcutaneous tissue disorders- Pruritus
3.2Postmarketing Experience
The following adverse reactions have been identified during post approval use of Teflaro in adult patients. Because these adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Agranulocytosis, leukopenia, eosinophilic pneumonia.
4OVERDOSAGE
Teflaro overdosage has occurred in patients with renal impairment. Reactions have included neurological sequelae, including encephalopathy
In the event of overdose, Teflaro should be discontinued and general supportive treatment given. Ceftaroline can be removed by hemodialysis. In subjects with ESRD administered 400 mg of Teflaro, the mean total recovery of ceftaroline in the dialysate following a 4-hour hemodialysis session started 4 hours after dosing was 76.5 mg (21.6% of the dose). However, no information is available on the use of hemodialysis to treat overdosage
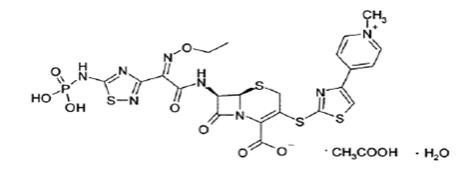
5DESCRIPTION
TEFLARO (ceftaroline fosamil) for injection, is a sterile, semi-synthetic, prodrug of the cephalosporin antibacterial class of beta-lactams (β-lactams) for intravenous use. Chemically, the prodrug, ceftaroline fosamil monoacetate monohydrate is (6
Figure 1: Chemical structure of ceftaroline fosamil
Teflaro vials contain either 600 mg or 400 mg of anhydrous ceftaroline fosamil (equivalent to 668 mg and 446 mg, respectively, of ceftaroline fosamil monoacetate monohydrate). The powder for injection is formulated from ceftaroline fosamil monoacetate monohydrate, a pale yellowish-white to light yellow sterile powder. Each vial of 400 mg of anhydrous ceftaroline fosamil includes inactive ingredient L-arginine 263.3 mg/vial as the solubilizer and alkalizing agent. Each vial of 600 mg of anhydrous ceftaroline fosamil includes inactive ingredient L-arginine 395.0 mg/vial as the solubilizer and alkalizing agent. All references to ceftaroline activity are expressed in terms of the prodrug, ceftaroline fosamil. The powder is constituted for IV injection
The pH of the constituted solution is pH 4.8 to 6.5.
6HOW SUPPLIED/STORAGE AND HANDLING
Teflaro (ceftaroline fosamil) for injection, a pale yellowish-white to light yellow sterile powder, is supplied in single-dose, clear glass vials containing:
- 600 mg - individual vial (NDC 0456-0600-01) and carton containing 10 vials (NDC 0456-0600-10)
- 400 mg - individual vial (NDC 0456-0400-01) and carton containing 10 vials (NDC 0456-0400-10)
Teflaro vials (unreconstituted) should be stored at 25ºC (77ºF); excursions permitted to 15-30ºC (59-86ºF) [see USP Controlled Room Temperature].
7PATIENT COUNSELING INFORMATION
- Advise patients that allergic reactions, including serious allergic reactions, could occur and that serious reactions require immediate treatment. They should inform their healthcare provider about any previous hypersensitivity reactions to Teflaro, other beta-lactams (including cephalosporins) or other allergens.
- Advise patients that neurological adverse reactions could occur with Teflaro use. Instruct patients or their caregivers to inform their healthcare provider at once of any neurological signs and symptoms, including encephalopathy (disturbance of consciousness including somnolence, lethargy, confusion, and slow thinking), and seizures for immediate treatment, dosage adjustment, or discontinuation of Teflaro.
- Patients should be counseled that antibacterial drugs including Teflaro should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Teflaro is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Teflaro or other antibacterial drugs in the future.
- Advise patients that diarrhea is a common problem caused by antibacterial drugs, including Teflaro and usually resolves when the drug is discontinued. Sometimes, frequent watery or bloody diarrhea may occur and may be a sign of a more serious intestinal infection. If severe watery or bloody diarrhea develops, patients should contact their healthcare provider.
Distributed by:
AbbVie Inc.
North Chicago, IL 60064
AbbVie Inc.
North Chicago, IL 60064
TEFLARO and its design are trademarks of Allergan Sales, LLC, an AbbVie company.
© 2024 AbbVie. All rights reserved.
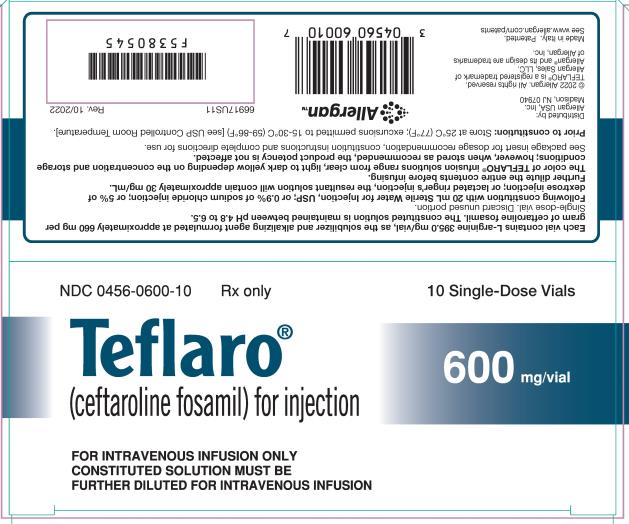
8PRINCIPAL DISPLAY PANEL
NDC 0456-0400-10
Rx only
Teflaro®
(ceftaroline fosamil) for injection
10 Single-Dose Vials
400 mg/vial
FOR INTRAVENOUS INFUSION ONLY
CONSTITUTED SOLUTION MUST BE
FURTHER DILUTED FOR INTRAVENOUS INFUSION
abbvie
Rx only
Teflaro®
(ceftaroline fosamil) for injection
10 Single-Dose Vials
400 mg/vial
FOR INTRAVENOUS INFUSION ONLY
CONSTITUTED SOLUTION MUST BE
FURTHER DILUTED FOR INTRAVENOUS INFUSION
abbvie
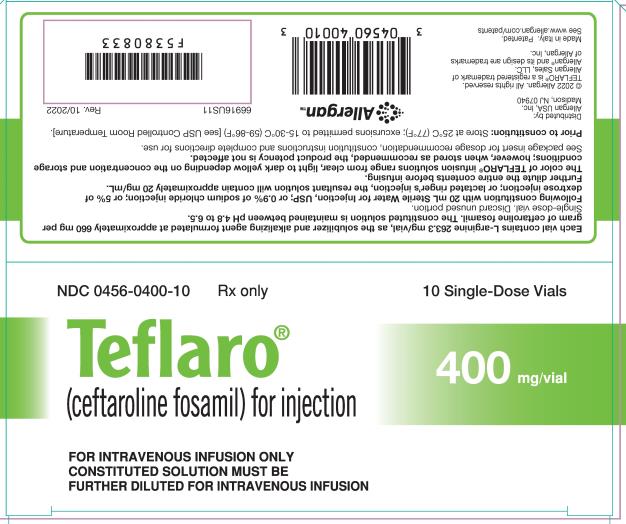
9PRINCIPAL DISPLAY PANEL
NDC 0456-0600-10
Rx only
Teflaro®
(ceftaroline fosamil) for injection
10 Single-Dose Vials
600 mg/vial
FOR INTRAVENOUS INFUSION ONLY
CONSTITUTED SOLUTION MUST BE
FURTHER DILUTED FOR INTRAVENOUS INFUSION
abbvie
Rx only
Teflaro®
(ceftaroline fosamil) for injection
10 Single-Dose Vials
600 mg/vial
FOR INTRAVENOUS INFUSION ONLY
CONSTITUTED SOLUTION MUST BE
FURTHER DILUTED FOR INTRAVENOUS INFUSION
abbvie