ARIPiprazole
What is Abilify Asimtufii (ARIPiprazole)?
Related Clinical Trials
Summary: This study will investigate neural markers in the cerebellar-cortical circuitry that predict treatment response to antipsychotics in first-episode psychosis. A total of 120 patients diagnosed with a first-episode psychotic disorder will be recruited. All subjects receive naturalistic treatment with either risperidone or aripiprazole for 12 weeks, with dosage and titration followed by standard clin...
Summary: The effectiveness and efficacy of the combination of pharmacotherapy with the two new recovery-oriented programs, RECOVERYTRSGR for patients with treatment- resistant schizophrenia and RECOVERYTRSBDGR for patients with treatment- resistant bipolar disorder.
Summary: This project aims to investigate the effectiveness of existing common antidepressants and to provide new evidence for depressed children and adolescents who are not responding to their first treatment.
Related Latest Advances
Brand Information
- for the treatment of schizophrenia in adults
- for maintenance monotherapy treatment of bipolar I disorder in adults
- Increased Mortality in Elderly Patients with Dementia - Related Psychosis Use
- Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients with Dementia-Related Psychosis
- Neuroleptic Malignant Syndrome
- Tardive Dyskinesia
- Metabolic Changes
- Pathological Gambling and Other Compulsive Behaviors
- Orthostatic Hypotension and Syncope
- Falls
- Leukopenia, Neutropenia, and Agranulocytosis
- Seizures
- Potential for Cognitive and Motor Impairment
- Body Temperature Regulation
- Dysphagia
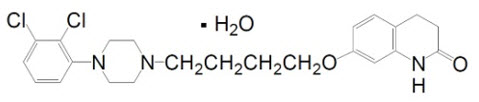
- Read the complete instructions for preparation and administration below before administering ABILIFY ASIMTUFII.
- To be prepared and administered only by a healthcare professional once every two months
- ABILIFY ASIMTUFII pre-filled syringe is
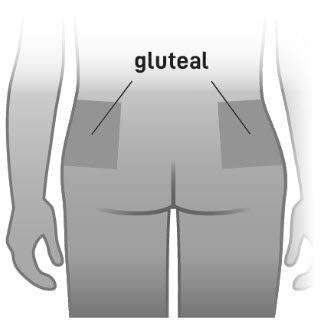
- For gluteal intramuscular injection only. Do not administer by any other route.
- Prior to administration, visually inspect ABILIFY ASIMTUFII pre-filled syringe for particulate matter and discoloration. The suspension should appear to be a uniform, homogeneous suspension that is opaque and milky-white in color. Do not use ABILIFY ASIMTUFII pre-filled syringe if the suspension is discolored, or particulate matter is present.
- One sterile 1 ½ inch 22 gauge needle (in black packaging)
- One sterile 2 inch 21 gauge needle (in green packaging)
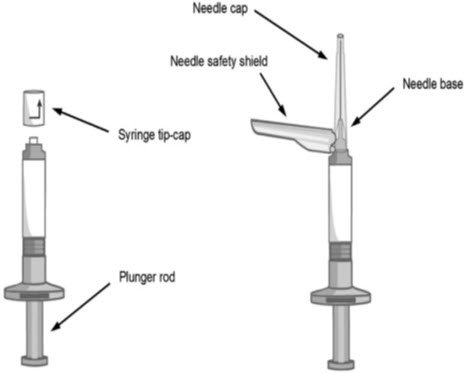
- Remove the ABILIFY ASIMTUFII pre-filled syringe from the package.
- Tap the syringe on your hand at least 10 (ten) times (Figure 1).
- After tapping, shake the syringe vigorously for at least 10 (ten) seconds until the medication is uniform (Figure 2).
- For non-obese patients - 22-gauge, 1.5-inch (38 mm) safety needle with needle protection device (needle in black packaging)
- For obese patients - 21-gauge, 2-inch (51 mm) safety needle with needle protection device (needle in green packaging)
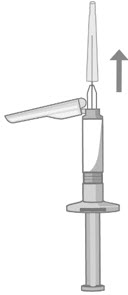
- Twist and pull off the pre-filled syringe tip-cap (Figure 3).
- While holding the base of the needle, ensure the needle is firmly seated on the safety device with a push. Gently twist clockwise until SECURELY fitted (Figure 3).

- When you are ready to administer the injection of ABILIFY ASIMTUFII, hold the pre-filled syringe upright and remove the needle-cap straight up (Figure 4). Do not twist the needle-cap, as this may loosen the needle from the syringe.
- Slowly advance the plunger rod upward to expel the air and until the suspension fills needle base (Figure 5).
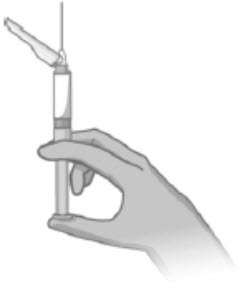
- Slowly inject the entire contents of the pre-filled syringe intramuscularly into the gluteal muscle of the patient (Figure 6).
- After the injection, press the safety shield on a hard surface to cover and lock shield over the needle (Figure 7 and 8)
- Immediately discard used syringe and the unused needle in an approved sharps container (Figure 9).
- The unused needle should not be saved for future use.




