Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Treatment of Schizophrenia in Adults
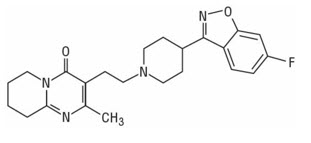
UZEDY is to be administered subcutaneously
The data described in this section are derived from a clinical trial database consisting of 9,803 patients exposed to one or more doses of oral risperidone for the treatment of schizophrenia and other psychiatric disorders. Of these 9,803 patients, 2,687 were patients who received oral risperidone while participating in double-blind, placebo-controlled trials. The conditions and duration of treatment with oral risperidone varied greatly and included (in overlapping categories) double-blind, fixed- and flexible-dose, placebo- or active-controlled studies and open-label phases of studies, inpatients and outpatients, and short-term (up to 12 weeks) and longer-term (up to 3 years) exposures. Safety was assessed by collecting adverse reactions and performing physical examinations, vital signs, body weights, laboratory analyses, and ECGs.
Injection site reactions for UZEDY presented in this section (see “Injection Site Reactions with UZEDY” below) are based on a randomized withdrawal study in patients with schizophrenia consisting of a 12-week open-label oral risperidone (2 mg to 5 mg) stabilization phase, followed by a placebo-controlled phase in which patients were randomized to UZEDY (once monthly or once every 2 months) or placebo for a variable time until impending relapse or study completion
The safety of UZEDY was evaluated in a total of 740 adult patients with schizophrenia who received at least 1 dose of UZEDY during the clinical development program. A total of 351 patients were exposed to UZEDY for at least 6 months, of which 221 patients were exposed to UZEDY for at least 12 months, which included 112 patients exposed to once monthly and 109 patients to once every 2 months dosing regimens. In addition, 32 patients were exposed to UZEDY for at least 24 months.
Treatment of Bipolar Disorder in Adults
UZEDY is to be administered subcutaneously
The safety of UZEDY as monotherapy or as adjunctive therapy to lithium or valproate for the maintenance treatment of Bipolar I Disorder in adults is based on adequate and well-controlled studies of another risperidone long-acting injection given once every 2 weeks. The results of those adequate and well-controlled studies are presented below.
Safety data are presented from a trial assessing the efficacy and safety of another risperidone long-acting injection, given once every 2 weeks, when administered as monotherapy for maintenance treatment in patients with bipolar I disorder.
Safety data are also presented from a trial assessing the efficacy and safety of another risperidone long-acting injection, given once every 2 weeks, administered as adjunctive maintenance treatment in patients with bipolar disorder.
Adverse Reactions in Studies with Oral Risperidone
The most common adverse reactions in clinical trials of oral risperidone (>5% and twice placebo) were parkinsonism, akathisia, dystonia, tremor, sedation, dizziness, anxiety, blurred vision, nausea, vomiting, upper abdominal pain, stomach discomfort, dyspepsia, diarrhea, salivary hypersecretion, constipation, dry mouth, increased appetite, increased weight, fatigue, rash, nasal congestion, upper respiratory tract infection, nasopharyngitis, and pharyngolaryngeal pain.
Commonly-Observed Adverse Reactions in Double-Blind, Placebo-Controlled Clinical Trials – Adult Patients with Schizophrenia Treated with Oral Risperidone
Table 7 lists the adverse reactions reported in 2% or more of oral risperidone-treated adult patients with schizophrenia in three 4- to 8-week, double-blind, placebo-controlled trials.
* Parkinsonism includes extrapyramidal disorder, musculoskeletal stiffness, parkinsonism, cogwheel rigidity, akinesia, bradykinesia, hypokinesia, masked facies, muscle rigidity, and Parkinson’s disease. Akathisia includes akathisia and restlessness. Dystonia includes dystonia, muscle spasms, muscle contractions involuntary, muscle contracture, oculogyration, tongue paralysis. Tremor includes tremor and parkinsonian rest tremor.
Adverse Reactions in Studies with Another Long-Acting Injection Risperidone, Given Once Every 2 Weeks
The most common adverse reactions in the double-blind, placebo-controlled periods of the bipolar disorder trials of another risperidone long-acting injection were weight increased (5% in the monotherapy trial) and tremor and parkinsonism (≥ 10% in the adjunctive treatment trial).
Commonly-Observed Adverse Reactions in Double-Blind, Placebo-Controlled Clinical Trials – Adult Patients with Bipolar Disorder Treated with Another Risperidone Long-Acting Injection Given Once Every 2 Weeks
Table 8 lists the treatment-emergent adverse reactions reported in 2% or more of patients treated with another risperidone long-acting injection, given once every 2 weeks, in the 24-month double-blind, placebo-controlled treatment period of the trial assessing the efficacy and safety of another risperidone long-acting injection when administered as monotherapy for maintenance treatment in patients with bipolar I disorder.
a The data presented are from a study with another risperidone long-acting injection that was administered once every 2 weeks, intramuscularly. UZEDY is to be administered subcutaneously.
Table 9 lists the treatment-emergent adverse reactions reported in 4% or more of patients in the 52-week double-blind, placebo-controlled treatment phase of a trial assessing the efficacy and safety of another Risperidone Long-Acting Injection, given once every 2 weeks, when administered as adjunctive maintenance treatment in patients with bipolar disorder.
a The data presented are from a study with another risperidone long-acting injection that was administered once every 2 weeks, intramuscularly. UZEDY is to be administered subcutaneously.
b Patients received double-blind treatment with another risperidone long-acting injection, given once every 2 weeks, or placebo in addition to continuing their treatment as usual, which included mood stabilizers, antidepressants, and/or anxiolytics.
c Parkinsonism includes muscle rigidity, hypokinesia, cogwheel rigidity, and bradykinesia.
d Dyskinesia includes muscle twitching and dyskinesia.
e Sedation includes sedation and somnolence.
Other Adverse Reactions Observed During the Clinical Trial Evaluations of Oral Risperidone
The following is a list of additional adverse drug reactions that have been reported during the clinical trial evaluation of oral risperidone:
Blood and Lymphatic System Disorders: anemia, granulocytopenia, neutropenia
Cardiac Disorders: sinus bradycardia, sinus tachycardia, atrioventricular block first degree, bundle branch block left, bundle branch block right, atrioventricular block
Ear and Labyrinth Disorders: ear pain, tinnitus
Endocrine Disorders:hyperprolactinemia
Eye Disorders: ocular hyperemia, eye discharge, conjunctivitis, eye rolling, eyelid edema, eye swelling, eyelid margin crusting, dry eye, lacrimation increased, photophobia, glaucoma, visual acuity reduced
Gastrointestinal Disorders: dysphagia, fecaloma, fecal incontinence, gastritis, lip swelling, cheilitis, aptyalism
General Disorders: edema peripheral, thirst, gait disturbance, chest discomfort, chest pain, influenza-like illness, pitting edema, edema, chills, sluggishness, malaise, face edema, discomfort, generalized edema, drug withdrawal syndrome, peripheral coldness, feeling abnormal
Immune System Disorders: drug hypersensitivity
Infections and Infestations: pneumonia, influenza, ear infection, viral infection, pharyngitis, tonsillitis, bronchitis, eye infection, localized infection, cystitis, cellulitis, otitis media, onychomycosis, acarodermatitis, bronchopneumonia, respiratory tract infection, tracheobronchitis, otitis media chronic
Investigations: body temperature increased, blood prolactin increased, alanine aminotransferase increased, electrocardiogram abnormal, eosinophil count increased, white blood cell count decreased, blood glucose increased, hemoglobin decreased, hematocrit decreased, body temperature decreased, blood pressure decreased, transaminases increased
Metabolism and Nutrition Disorders: decreased appetite, polydipsia, anorexia
Musculoskeletal, Connective Tissue, and Bone Disorders: joint swelling, joint stiffness, musculoskeletal chest pain, posture abnormal, myalgia, neck pain, muscular weakness, muscle rigidity, rhabdomyolysis, torticollis
Nervous System Disorders: balance disorder, disturbance in attention, dysarthria, unresponsive to stimuli, depressed level of consciousness, movement disorder, transient ischemic attack, coordination abnormal, cerebrovascular accident, speech disorder, syncope, loss of consciousness, hypoesthesia, tardive dyskinesia, cerebral ischemia, cerebrovascular disorder, neuroleptic malignant syndrome, diabetic coma, head titubation
Psychiatric Disorders: agitation, blunted affect, confusional state, middle insomnia, nervousness, sleep disorder, listlessness, libido decreased, anorgasmia
Renal and Urinary Disorders: enuresis, dysuria, pollakiuria, urinary incontinence
Reproductive System and Breast Disorders: menstruation irregular, amenorrhea, gynecomastia, galactorrhea, vaginal discharge, menstrual disorder, erectile dysfunction, retrograde ejaculation, ejaculation disorder, sexual dysfunction, breast enlargement
Respiratory, Thoracic, and Mediastinal Disorders: wheezing, pneumonia aspiration, sinus congestion, dysphonia, productive cough, pulmonary congestion, respiratory tract congestion, rales, respiratory disorder, hyperventilation, nasal edema
Skin and Subcutaneous Tissue Disorders: erythema, skin discoloration, skin lesion, pruritus, skin disorder, rash erythematous, rash papular, acne, hyperkeratosis, seborrheic dermatitis, rash generalized, rash maculopapular
Vascular Disorders: hypotension, flushing
Additional Adverse Reactions Reported With Another Risperidone Long-Acting Injection, Given Once Every 2 Weeks
The following is a list of additional adverse reactions that have been reported during the premarketing evaluation of another risperidone long-acting injection, given once every 2 weeks, regardless of frequency of occurrence:
Cardiac Disorders: bradycardia, tachycardia, palpitations
Ear and Labyrinth Disorders: vertigo
Eye Disorders: blepharospasm
Gastrointestinal Disorders: toothache, tongue spasm, diarrhea, vomiting, abdominal pain upper, abdominal pain, stomach discomfort
General Disorders and Administration Site Conditions: pain, injection site pain, induration, injection site induration, injection site swelling, injection site reaction
Infections and Infestations: lower respiratory tract infection, infection, gastroenteritis, subcutaneous abscess, nasopharyngitis, urinary tract infection, rhinitis, sinusitis
Injury and Poisoning: fall, procedural pain
Investigations: weight decreased, gamma-glutamyltransferase increased, hepatic enzyme increased, aspartate aminotransferase increased, electrocardiogram QT prolonged, glucose urine present
Musculoskeletal, Connective Tissue, and Bone Disorders: buttock pain, back pain
Nervous System Disorders: convulsion, paresthesia, dystonia, drooling, dizziness postural, akinesia, hypokinesia
Psychiatric Disorders: depression, insomnia, anxiety, initial insomnia
Reproductive System and Breast Disorders: oligomenorrhea, breast discomfort, menstruation delayed, ejaculation delayed
Respiratory, Thoracic, and Mediastinal Disorders: nasal congestion, pharyngolaryngeal pain, dyspnea, rhinorrhea
Skin and Subcutaneous Tissue Disorders: eczema, rash, pruritis generalized
Vascular Disorders: hypertension, orthostatic hypotension
Discontinuations Due to Adverse Drug Reactions with Oral Risperidone
Schizophrenia
Approximately 7% (39/564) of oral risperidone-treated patients in double-blind, placebo-controlled trials discontinued treatment due to an adverse reaction, compared with 4% (10/225) who were receiving placebo. The adverse reactions associated with discontinuation in 2 or more oral risperidone-treated patients were:
Discontinuation for extrapyramidal symptoms (including Parkinsonism, akathisia, dystonia, and tardive dyskinesia) was 1% in placebo-treated patients, and 3.4% in active control-treated patients in a double-blind, placebo- and active-controlled trial.
Discontinuations Due to Adverse Drug Reactions with Another Long-Acting Injection Risperidone, Given Once Every 2 Weeks
Bipolar Disorder
In a 24-month double-blind, placebo-controlled treatment period of the trial assessing the efficacy and safety of another risperidone long-acting injection, given once every 2 weeks, monotherapy for maintenance treatment in patients with bipolar I disorder, 1 (0.6%) of 154 patients treated with another risperidone long-acting injection, given once every 2 weeks, discontinued due to an adverse reaction (hyperglycemia).
In the 52-week double-blind phase of the placebo-controlled trial in which patients treated with another risperidone long-acting injection, given once every 2 weeks, was administered as adjunctive therapy to patients with bipolar disorder in addition to continuing with their usual treatment, approximately 4% (3/72) of patients treated with another risperidone long-acting injection, given once every 2 weeks, discontinued treatment due to an adverse event, compared with 1.5% (1/67) of placebo-treated patients. Adverse reactions associated with discontinuation in patients treated with another risperidone long-acting injection, given once every 2 weeks, were hypokinesia (one patient) and tardive dyskinesia (one patient).
Dose Dependency of Adverse Reactions in Clinical Trials of Oral Risperidone
Extrapyramidal Symptoms
Data from two fixed-dose trials in adults with schizophrenia provided evidence of dose-relatedness for extrapyramidal symptoms associated with oral risperidone treatment. Two methods were used to measure extrapyramidal symptoms (EPS) in an 8-week trial comparing 4 fixed doses of oral risperidone (2, 6, 10, and 16 mg/day), including (1) a Parkinsonism score (mean change from baseline) from the Extrapyramidal Symptom Rating Scale, and (2) incidence of spontaneous complaints of EPS:
Similar methods were used to measure extrapyramidal symptoms (EPS) in an 8-week trial comparing 5 fixed doses of oral risperidone (1, 4, 8, 12, and 16 mg/day):
Changes in Body Weight
Weight gain was observed in short-term, controlled trials and longer-term uncontrolled studies in adults
Dystonia
Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Other Adverse Reactions
Adverse reaction data elicited by a checklist for side effects from a large study comparing 5 fixed doses of oral risperidone (1, 4, 8, 12, and 16 mg/day) were explored for dose-relatedness of adverse events. A Cochran-Armitage Test for trend in these data revealed a positive trend (p<0.05) for the following adverse reactions: somnolence, vision abnormal, dizziness, palpitations, weight increase, erectile dysfunction, ejaculation disorder, sexual function abnormal, fatigue, and skin discoloration.
Changes in ECG
Between-group comparisons for pooled placebo-controlled trials of oral risperidone in adults revealed no statistically significant differences between oral risperidone and placebo in mean changes from baseline in ECG parameters, including QT, QTc, and PR intervals, and heart rate. When all oral risperidone doses were pooled from randomized controlled trials in several indications, there was a mean increase in heart rate of 1 beat per minute compared to no change for placebo patients. In short-term schizophrenia trials, higher doses of oral risperidone were associated with a higher mean increase in heart rate compared to placebo (4 to 6 beats per minute).
The ECGs of 227 patients with Bipolar I Disorder were evaluated in the 24-month double-blind, placebo-controlled period. There were no clinically relevant differences in QTc intervals (using Fridericia's and linear correction factors) during treatment with another risperidone long-acting injection, given once every 2 weeks, compared to placebo.
The ECGs of 85 patients with bipolar disorder were evaluated in the 52-week double-blind, placebo-controlled trial. There were no statistically significant differences in QTc intervals (using Fridericia's and linear correction factors) during adjunctive treatment with either another risperidone long-acting injection (25 mg, 37.5 mg, or 50 mg), given once every 2 weeks, or placebo in addition to treatment as usual.
Injection Site Reactions with UZEDY
Local tolerability assessments were administered to patients who reported injection site adverse reactions in a randomized withdrawal study with UZEDY in adult patients with schizophrenia. The injection site was assessed by appropriately trained personnel throughout the clinical development program.
All injection site reactions (nodule, pruritus, erythema, mass, and swelling) were mild to moderate in severity with the exception of 1 case of severe pruritus which resolved after 6 days. Injection site reactions were reported in 22 patients (13%) in the placebo group, 36 patients (20%) in the UZEDY once monthly group, and 37 patients (21%) in the UZEDY once every 2 months group. The most common injection site reactions were: nodule (7% in each UZEDY-treated group and 3% in the placebo group) and pruritus (5% and 3% in the UZEDY-treated once monthly and once every 2 months groups, respectively, and 2% in the placebo group).