Atherosclerosis Treatments
Find Atherosclerosis Treatments
Xarelto
What is Xarelto (Rivaroxaban)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: Through a prospective, single-center, randomized controlled trial, we aim to determine the thromboprophylactic efficacy of rivaroxaban in patients with idiopathic membranous nephropathy (IMN). IMN patients at high risk of thrombosis and low risk of bleeding will be enrolled and randomly assigned to a rivaroxaban group or a control group (receiving warfarin). Prophylactic anticoagulation will be ad...
Summary: This study is a prospective, multicenter, randomized controlled trial of an interventional strategy using the ClotTriever System to achieve and maintain vessel patency (ClotTriever Intervention Arm) versus conservative medical management using anticoagulation therapy alone (Conservative Medical Management Arm) in the treatment of subjects with symptomatic unilateral iliofemoral DVT. The study will...
Summary: This study is a prospective, multicenter, randomized controlled trial of the FlowTriever System plus anticoagulation compared to anticoagulation alone for intermediate-risk acute PE.
Related Latest Advances
Brand Information
- use of indwelling epidural catheters
- concomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants
- a history of traumatic or repeated epidural or spinal punctures
- a history of spinal deformity or spinal surgery
- optimal timing between the administration of XARELTO and neuraxial procedures is not known
- 2.5 mg tablets: Round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side
- 10 mg tablets: Round, light red, biconvex and film-coated with a triangle pointing down above a "10" marked on one side and "Xa" on the other side
- 15 mg tablets: Round, red, biconvex, and film-coated with a triangle pointing down above a "15" marked on one side and "Xa" on the other side
- 20 mg tablets: Triangle-shaped, dark red, and film-coated with a triangle pointing down above a "20" marked on one side and "Xa" on the other side
- For oral suspension: white to off-white granules; once reconstituted, provide flavored white to off-white opaque liquid with a concentration of 1 mg/mL.
- active pathological bleeding
- severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions)
- Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation
- Bleeding Risk
- Spinal/Epidural Hematoma
- 2.5 mg tablets are round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
- 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed:
- 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
- 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
- Starter Pack for treatment of deep vein thrombosis and treatment of pulmonary embolism:
- Advise patients to take XARELTO only as directed.
- Remind patients to not discontinue XARELTO without first talking to their healthcare professional.
- Advise patients with atrial fibrillation to take XARELTO once daily with the evening meal.
- Advise patients for initial treatment of DVT and/or PE to take XARELTO 15 mg or 20 mg tablets with food at approximately the same time every day
- Advise patients who are at a continued risk of recurrent DVT and/or PE after at least 6 months of initial treatment, to take XARELTO 10 mg once daily with or without food
- Advise patients who cannot swallow the tablet whole to crush XARELTO and combine with a small amount of applesauce followed by food
- For patients requiring an NG tube or gastric feeding tube, instruct the patient or caregiver to crush the XARELTO tablet and mix it with a small amount of water before administering via the tube
- If a dose is missed, advise the patient according to the instructions in the Full Prescribing Information based on their dosing schedule
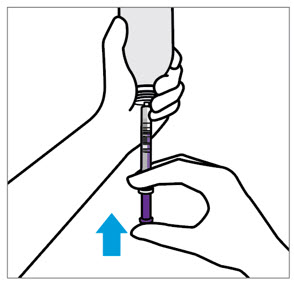
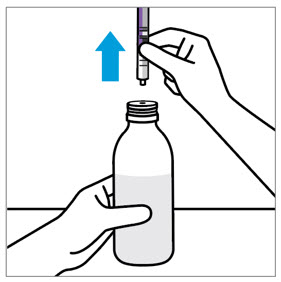
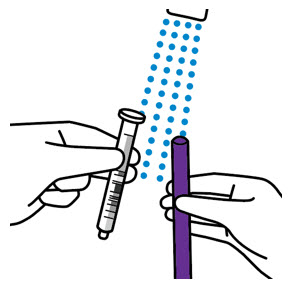
- The adult caregiver should administer the dose. Advise caregivers to use the syringes provided in the original carton.
- Advise the caregiver whether the dose needs to be taken with food or not
- Advise the caregiver the tablet must not be split in an attempt to provide a fraction of a tablet dose
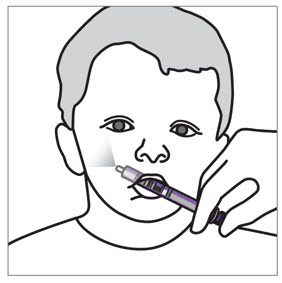
- If a child vomits or spits up the dose within 30 minutes after receiving the dose, a new dose should be given. However, if the child vomits more than 30 minutes after the dose is taken, the dose should not be re-administered and the next dose should be taken as scheduled. If a child vomits or spits up the dose repeatedly, the caregiver should contact the child's doctor right away
- For children who are unable to swallow whole tablets, XARELTO oral suspension may be used.
- If a dose is missed, advise the patient according to the instructions in the Full Prescribing Information based on their dosing schedule
- Advise patients to report any unusual bleeding or bruising to their physician. Inform patients that it might take them longer than usual to stop bleeding, and that they may bruise and/or bleed more easily when they are treated with XARELTO
- If patients have had neuraxial anesthesia or spinal puncture, and particularly, if they are taking concomitant NSAIDs or platelet inhibitors, advise patients to watch for signs and symptoms of spinal or epidural hematoma, such as back pain, tingling, numbness (especially in the lower limbs), muscle weakness, and stool or urine incontinence. If any of these symptoms occur, advise the patient to contact his or her physician immediately
- Advise patients to inform their physician immediately if they become pregnant or intend to become pregnant during treatment with XARELTO
- Advise pregnant women receiving XARELTO to immediately report to their physician any bleeding or symptoms of blood loss
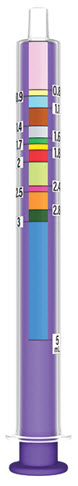
- XARELTO suspension is for oral use only.
- Give XARELTO to your child exactly as prescribed by your doctor. The adult caregiver should give the dose. If you have questions, contact your doctor or pharmacist for more information on giving a dose.
- Only use the oral dosing syringe provided with XARELTO oral suspension. Contact your doctor or pharmacist if the oral dosing syringe is missing, lost or damaged.
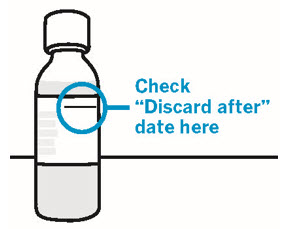
 Storage information
Storage information
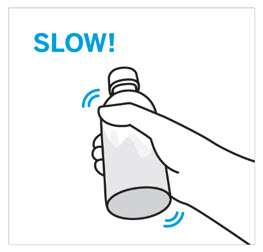
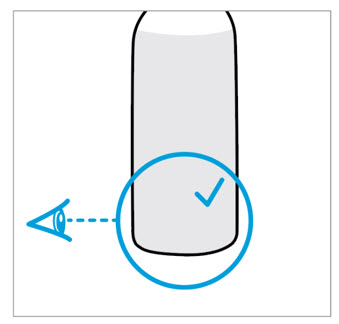
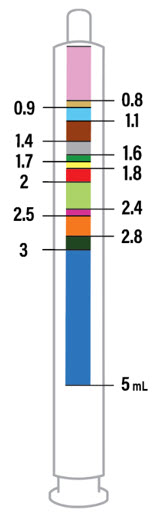
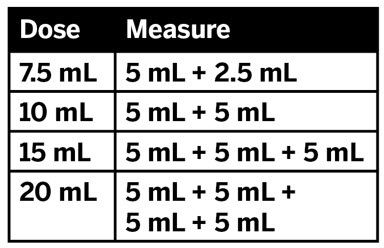
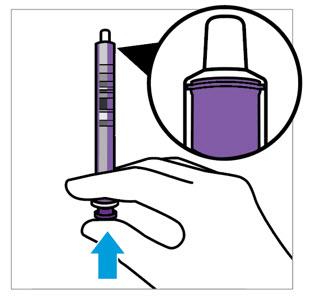
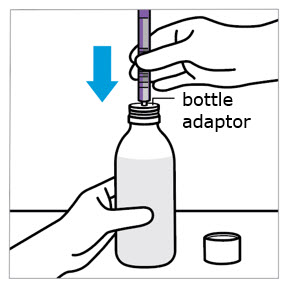
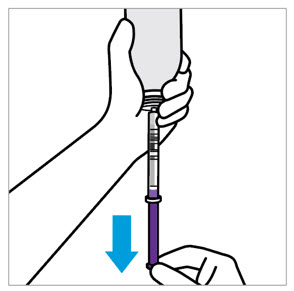
 CAUTION:
CAUTION: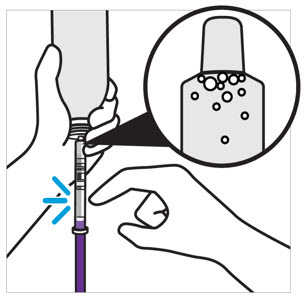
- Throw the XARELTO bottle away in your household trash.
- Throw away any used oral dosing syringe with the opening of a new XARELTO bottle.
- Do not pour XARELTO suspension down the drain (for example: sink, toilet, shower or tub).
- Do not recycle the bottle.





- Must reconstitute before
- Counsel caregiver on proper use.





