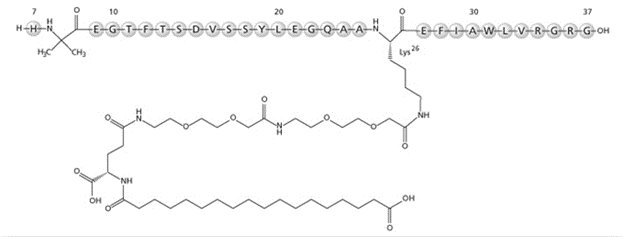
Semaglutide
What is Ozempic (Semaglutide)?
Approved To Treat
Related Clinical Trials
Summary: This multicenter, randomized, double-blind, placebo- and open-label active comparator-controlled, parallel-group, dose-range-finding, Phase II study aims to evaluate the efficacy, tolerability, and safety of RO7795081 for glycemic control in adult participants with Type 2 diabetes mellitus (T2D).
Summary: The purpose of this study is to evaluate the efficacy and safety of ofirnoflast in combination with semaglutide in comparison to semaglutide alone in obese participants with T2DM.
Summary: This study will evaluate the effects of oral semaglutide in combination with topical corticosteroid/calcipotriol on clinical outcomes and metabolic inflammation in patients with plaque psoriasis and overweight/obesity and/or type 2 diabetes mellitus. A total of 62 participants will be randomized to receive either semaglutide plus topical corticosteroid/calcipotriol or placebo plus topical corticos...
Related Latest Advances
Brand Information
- In rodents, semaglutide causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures. It is unknown whether OZEMPIC causes thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), in humans as human relevance of semaglutide-induced rodent thyroid C-cell tumors has not been determined
- OZEMPIC is contraindicated in patients with a personal or family history of MTC or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, non-fatal myocardial infarction or non-fatal stroke) in adults with type 2 diabetes mellitus and established cardiovascular disease.
- OZEMPIC has not been studied in patients with a history of pancreatitis. Consider other antidiabetic therapies in patients with a history of pancreatitis
- OZEMPIC is not indicated for use in patients with type 1 diabetes mellitus.
- A personal or family history of MTC or in patients with MEN 2
- A serious hypersensitivity reaction to semaglutide or to any of the excipients in OZEMPIC. Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported with OZEMPIC
- In addition to the reactions in Table 1, the following gastrointestinal adverse reactions with a frequency of <5% were associated with OZEMPIC (frequencies listed, respectively, as: placebo; 0.5 mg; 1 mg): dyspepsia (1.9%, 3.5%, 2.7%), eructation (0%, 2.7%, 1.1%), flatulence (0.8%, 0.4%, 1.5%), gastroesophageal reflux disease (0%, 1.9%, 1.5%), and gastritis (0.8%, 0.8%, 0.4%).
- This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 03/2022





