Valtrex
What is Valtrex (ValACYclovir)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: Phase 2a, open-label, multi-center study evaluating biomarkers and biodistribution of aglatimagene besadenovec plus valacyclovir in men with localized, favorable intermediate-risk prostate cancer who are planning to receive external beam radiation therapy (EBRT).
Summary: Herpes Zoster (HZ), caused by the Varicella-Zoster Virus (VZV), is a common infection characterized by localized pain and blistering, with higher incidence in females. HZ affects patients' physical health, mental well-being, and quality of life. The immune system is critical in HZ pathogenesis. Studies show acupuncture relieves pain, modulates immunity, and may lower postherpetic neuralgia (PHN) r...
Summary: To learn if the combination of pirtobrutinib (also called LOXO-305), venetoclax, and obinutuzumab is safe and effective when given to patients with chronic lymphocytic leukemia (CLL) or Richter transformation (RT) who have not previously received treatment.
Related Latest Advances
Brand Information
- VALTREX may be given without regard to meals.
- Valacyclovir oral suspension (25 mg/mL or 50 mg/mL) may be prepared extemporaneously from 500-mg VALTREX tablets for use in pediatric patients for whom a solid dosage form is not appropriate
- Prepare SSV according to the USP-NF.
- Using a pestle and mortar, grind the required number of VALTREX 500-mg tablets until a fine powder is produced (5 VALTREX tablets for 25-mg/mL suspension; 10 VALTREX tablets for 50-mg/mL suspension).
- Gradually add approximately 5-mL aliquots of SSV to the mortar and triturate the powder until a paste has been produced. Ensure that the powder has been adequately wetted.
- Continue to add approximately 5-mL aliquots of SSV to the mortar, mixing thoroughly between additions, until a concentrated suspension is produced, to a minimum total quantity of 20 mL SSV and a maximum total quantity of 40 mL SSV for both the 25-mg/mL and 50-mg/mL suspensions.
- Transfer the mixture to a suitable 100-mL measuring flask.
- Transfer the cherry flavor* to the mortar and dissolve in approximately 5 mL of SSV. Once dissolved, add to the measuring flask.
- Rinse the mortar at least 3 times with approximately 5-mL aliquots of SSV, transferring the rinsing to the measuring flask between additions.
- Make the suspension to volume (100 mL) with SSV and shake thoroughly to mix.
- Transfer the suspension to an amber glass medicine bottle with a child-resistant closure.
- The prepared suspension should be labeled with the following information “Shake well before using. Store suspension between 2°C to 8°C (36°F to 46°F) in a refrigerator. Discard after 28 days.”
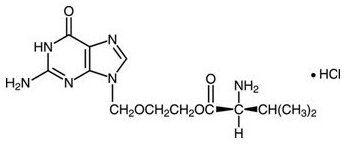
- 1-gram: Each blue, film‑coated, capsule‑shaped tablet, with a partial scorebar on both sides, printed with “VALTREX 1 gram” contains 1.112 grams of valacyclovir hydrochloride equivalent to 1 gram of the free base.
- Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome
- Acute Renal Failure
- Central Nervous System Effects
- Severe Cutaneous Adverse Reactions