Interstitial Keratitis Treatments
Find Interstitial Keratitis Treatments
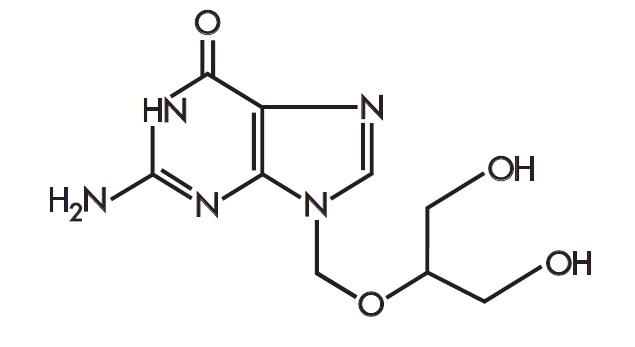
Zirgan
What is Zirgan (Ganciclovir)?
Approved To Treat
Top Global Experts
Related Clinical Trials
Summary: Over half of women in the US who are breastfeeding their infants take prescription drugs. You are being asked to participate in this study because you are breastfeeding your infant and are currently taking, as part of your medical care, at least one of the drugs we are studying. We are interested in studying drugs commonly prescribed to women who are breastfeeding so we can learn more about the am...
Summary: This is a Phase 1 single-arm open-label study of letermovir in neonates with symptomatic congenital Cytomegalovirus (CMV) disease. There will be two groups enrolled. Group 1 will be comprised of 4 subjects. Following documentation study inclusion and signing of informed consent, Group 1 subjects will receive one dose of oral letermovir (Study Day 0), using the dose bands. A full pharmacokinetics (...
Background: \- KSHV inflammatory cytokine syndrome (KICS) is a newly recognized disease caused by Kaposi sarcoma-associated herpesvirus (KSHV). This virus can cause cancer. People with KICS can have severe symptoms. They include fever, weight loss, and fluid in the legs or abdomen. People with KICS may also be at risk of getting other cancers associated with KSHV. These cancers include Kaposi sarcoma and lymp...
Related Latest Advances
Brand Information