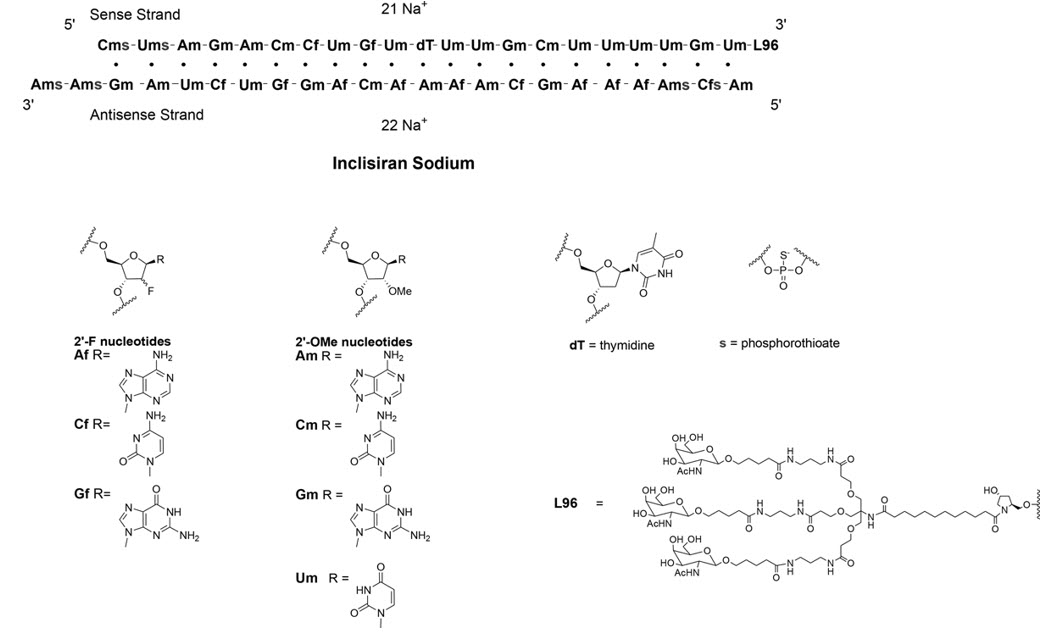
Adults with Primary Hypercholesterolemia or HeFH
The efficacy of LEQVIO was investigated in three randomized, double-blind, placebo-controlled trials that enrolled 3,660 adults with HeFH, clinical ASCVD, or increased risk for ASCVD, who were taking maximally tolerated statin therapy and who required additional LDL-C lowering. Demographics and baseline disease characteristics were balanced between the treatment arms in all trials.
Adults with Primary Hypercholesterolemia
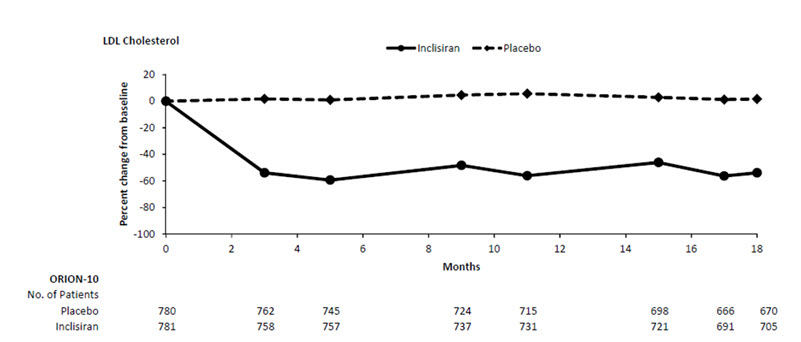
Trial 1 (ORION-10, NCT03399370) was a multicenter, double-blind, randomized, placebo-controlled 18-month trial in which 1,561 adults with ASCVD were randomized 1:1 to receive subcutaneous injections of either LEQVIO 284 mg (n = 781) or placebo (n = 780) on Day 1, Day 90, Day 270, and at Day 450. Patients were taking a maximally tolerated dose of statin with or without other lipid modifying therapy and required additional LDL-C reduction. Patients were stratified by current use of statins or other lipid-modifying therapies. Patients taking PCSK9 inhibitors were excluded from the trial.
The mean age at baseline was 66 years (range: 35 to 90 years), 60% were ≥65 years old, 31% were female, 86% were White, 13% were Black or African American, 1% were Asian, and 14% identified as Hispanic or Latino ethnicity. Forty-five percent (45%) of patients had diabetes at baseline. The mean baseline LDL-C was 105 mg/dL. At the time of randomization, 89% of patients were receiving statin therapy and 69% were receiving high-intensity statin therapy.
The primary efficacy outcome measure in Trial 1 was the percent change from baseline to Day 510 in LDL-C. The difference between the LEQVIO and placebo groups in mean percentage change in LDL-C from baseline to Day 510 was -52% (95% CI: -56%, -49%; p < 0.0001). For additional results, see Table 2 and Figure 1.
Figure 1: Mean Percent Change from Baseline in LDL-C Over 18 Months in Adults with Hypercholesterolemia and ASCVD on Maximally Tolerated Statin Therapy (Trial 1)
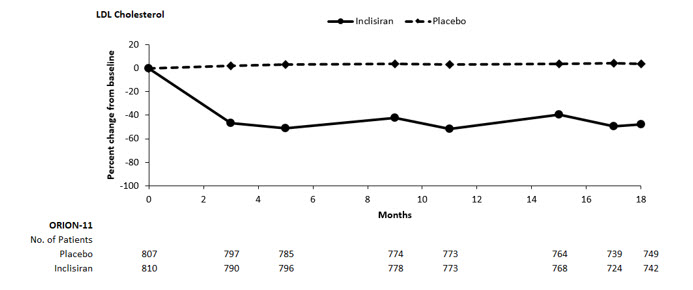
Trial 2 (ORION-11, NCT03400800) was a multicenter, double-blind, randomized, placebo-controlled 18-month trial in which 1,617 adults with ASCVD or increased risk for ASCVD were randomized 1:1 to receive subcutaneous injections of either LEQVIO 284 mg (n = 810) or placebo (n = 807) on Day 1, Day 90, Day 270, and Day 450. Patients were taking a maximally tolerated dose of statin with or without other lipid modifying therapy and required additional LDL-C reduction. Patients were stratified by country and by current use of statins or other lipid-modifying therapies. Patients taking PCSK9 inhibitors were excluded from the trial.
The mean age at baseline was 65 years (range: 20 to 88 years), 55% were ≥65 years old, 28% were female, 98% were White, 1% were Black or African American, and <1% were Asian; <1% identified as Hispanic or Latino ethnicity. Thirty-five percent (35%) of patients had diabetes at baseline. The mean baseline LDL-C was 105 mg/dL. At the time of randomization, 95% of patients were receiving statin therapy and 78% were receiving high-intensity statin therapy.
The primary efficacy outcome measure in Trial 2 was the percent change from baseline to Day 510 in LDL-C. The difference between the LEQVIO and placebo groups in mean percentage change in LDL-C from baseline to Day 510 was -50% (95% CI: -53%, -47%; p < 0.0001). For additional results, see Table 3 and Figure 2.
Figure 2: Mean Percent Change from Baseline in LDL-C Over 18 Months in Adults with Hypercholesterolemia and ASCVD or Increased Risk for ASCVD on Maximally Tolerated Statin Therapy (Trial 2)
In a pooled analysis of Trial 1 and Trial 2, the observed treatment effect was similar across predefined subgroups, such as sex, age, race, disease characteristics, geographic regions, presence of diabetes, body mass index, baseline LDL-C levels, and intensity of statin treatment.
Adults with HeFH
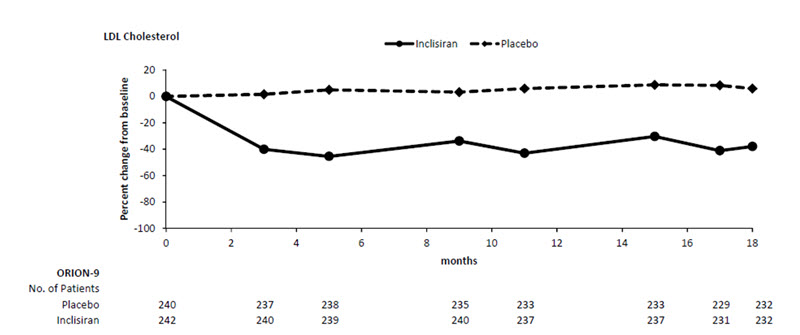
Trial 3 (ORION-9, NCT03397121) was a multicenter, double-blind, randomized, placebo-controlled 18-month trial in which 482 adults with HeFH were randomized 1:1 to receive subcutaneous injections of either LEQVIO 284 mg (n = 242) or placebo (n = 240) on Day 1, Day 90, Day 270, and at Day 450. Patients with HeFH were taking a maximally tolerated dose of statin with or without other lipid modifying therapy and required additional LDL-C reduction. The diagnosis of HeFH was made either by genotyping or clinical criteria using either the Simon Broome or WHO/Dutch Lipid Network criteria. Patients were stratified by country and by current use of statins or other lipid-modifying therapies. Patients taking PCSK9 inhibitors were excluded from the trial.
The mean age at baseline was 55 years (range: 21 to 80 years), 22% were ≥65 years old, 53% were female, 94% were White, 3% were Black or African American, and 3% were Asian; and 3% identified as Hispanic or Latino ethnicity. Ten percent (10%) of patients had diabetes at baseline. The mean baseline LDL-C was 153 mg/dL. At the time of randomization, 90% of patients were receiving statin therapy and 74% were receiving high-intensity statin therapy. Fifty-two percent (52%) of patients were treated with ezetimibe. The most commonly administered statins were atorvastatin and rosuvastatin.
The primary efficacy outcome measure in Trial 3 was the percent change from baseline to Day 510 in LDL-C. The difference between the LEQVIO and placebo groups in mean percentage change in LDL-C from baseline to Day 510 was -48% (95% CI: -54%, -42%; p < 0.0001). For additional results, see Table 4 and Figure 3.
Figure 3: Mean Percent Change from Baseline in LDL-C Over 18 Months in Adults with HeFH on Maximally Tolerated Statin Therapy (Trial 3)
Pediatric Patients with HeFH
Trial 4 (ORION-16, NCT04652726) was a 12-month randomized, double-blind, placebo-controlled trial in 141 pediatric patients aged 12 years and older with HeFH and elevated LDL-C. Patients were receiving maximally tolerated statin therapy with or without additional LDL-C-lowering therapies. The diagnosis of HeFH was made either by genetic testing or clinical criteria. Patients were randomized in a 2:1 ratio to receive subcutaneous injections of either LEQVIO 284 mg (n = 93) or placebo (n = 48) on Day 1, Day 90, and Day 270.
The mean age at baseline was 15 years (range: 12 to 17 years), 53% were female, 91% were White, 4% were Black or African American, 3% were Asian, and 3% were other races; 9% identified as Hispanic or Latino ethnicity. The mean LDL-C at baseline was 183 mg/dL; 93% of patients were taking statins and 23% were on ezetimibe.
The primary efficacy outcome measure in Trial 4 was the percent change from baseline to Day 330 in LDL-C. The difference between the LEQVIO and placebo groups in mean percentage change in LDL-C from baseline to Day 330 was -29% (95% CI: −36%, −21%; p < 0.0001). For additional results, see Table 5 and Figure 4.
Figure 4: Mean Percent Change from Baseline in LDL-C Over 12 Months in Pediatric Patients aged 12 Years and Older with HeFH (Trial 4)
Pediatric Patients with HoFH
Trial 5 (ORION-13, NCT04659863) was a 12-month randomized, double-blind, placebo-controlled trial in 13 pediatric patients aged 12 years and older with HoFH and elevated LDL-C. All patients were taking LDL-C-lowering therapies. Patients with a null (negative) variant in both low-density lipoprotein receptor (LDLR) alleles, who were considered unlikely to benefit from a reduction in PCSK9, were excluded. The diagnosis of HoFH was made by genetic testing. Patients were randomized in a 2:1 ratio to receive subcutaneous injections of either LEQVIO 284 mg (n = 9) or placebo (n = 4) on Day 1, Day 90, and Day 270.
The mean age at baseline was 15 years (range: 12 to 17 years), 69% were female, 85% were White, and 15% were Asian; 8% identified as Hispanic or Latino ethnicity. The mean LDL-C at baseline was 272 mg/dL; all patients were taking statins and 85% were on ezetimibe.
The primary efficacy outcome measure in Trial 5 was the percent change from baseline to Day 330 in LDL-C. The difference between the LEQVIO and placebo groups in mean percentage change in LDL-C from baseline to Day 330 was -33% (95% CI: −80%, 13%). For additional results, see Table 6 and Figure 5.
Figure 5: Mean Percent Change from Baseline in LDL-C Over 12 Months in Pediatric Patients aged 12 Years and Older with HoFH (Trial 5)